Synthesis
Diethyl 3-amino-1H-pyrrole-2,4-dicarboxylate (10.0 g, 44.2 mmol) and formamidine acetate (13.8 g, 133 mmol) were mixed in ethanol (100 mL) and heated to react for 20 hours at 105 °C. After the reaction was completed, the reaction mixture was filtered while hot, and the solid was collected and washed with ethanol. After the filtrate was cooled to room temperature, the solid was collected by filtration again and washed with ethanol. The two collected solids were combined, slurried with ether, filtered and dried under vacuum to afford ethyl 4,5-dihydro-4-oxo-1H-pyrrolo[3,2-d]pyrimidine-7-carboxylate (6.60 g, 31.9 mmol, 72% yield) as a light yellow solid, which could be used for subsequent reaction without further purification. The product was characterized by 1H NMR (500 MHz, DMSO-d6): δ 7.91 (s, 1H), 7.89 (s, 1H), 4.22 (q, J = 7.2 Hz, 2H), 3.17 (s, 2H), 1.27 (t, J = 7.2 Hz, 3H); LC/MS (ES+) m/z (M + H)+ = 208; HPLC Rt = 0.55 min, column N.
![ETHYL 4,5-DIHYDRO-4-OXO-1H-PYRROLO[3,2-D]PYRIMIDINE-7-CARBOXYLATE Structure](https://www.chemicalbook.com/CAS/GIF/853058-41-0.gif)
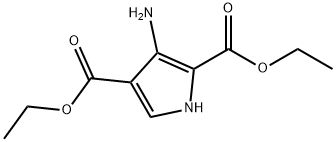
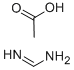
![ETHYL 4,5-DIHYDRO-4-OXO-1H-PYRROLO[3,2-D]PYRIMIDINE-7-CARBOXYLATE](/CAS/GIF/853058-41-0.gif)
