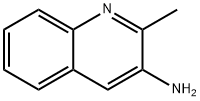
Synthese
2-Methyl-3-nitroquinoline (400 mg, 2.13 mmol) was used as starting material, which was dissolved in concentrated hydrochloric acid (8 mL) and heated to 50 °C. Subsequently, tin(II) chloride dihydrate (1.2 g, 5.3 mmol) was slowly added to the reaction system. The reaction temperature was maintained at 50 °C and the reaction mixture was continuously stirred overnight. After completion of the reaction, the mixture was diluted with distilled water (20 mL). The pH was adjusted to 9 with 5 N NaOH aqueous solution.The reaction mixture was cooled to 4 °C and extracted with ethyl acetate (2 × 30 mL). The organic phases were combined, washed with ice water (40 mL), dried over anhydrous Na2SO4, filtered and concentrated to give 3-amino-2-methylquinoline (270 mg, 80% yield) as a yellow solid. The product was characterized by 1H NMR (400 MHz, CDCl3): δ 7.84 (d, J = 8.4 Hz, 1H), 7.51 (dd, J = 1.2, 8.0 Hz, 1H), 7.39-7.30 (m, 2H), 7.16 (s, 1H), 3.77 (s, 2H), 2.56 (s, 3H).