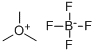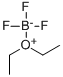The general procedure for the preparation of trimethoxonium tetrafluoroborate is as follows (adapted with minor modifications from TJ. Curphey, Org. Synth., Coll. vol. 6, p. 1019 (1988)):
1. add 80 mL of dichloromethane (DCM) to a 500 mL three-necked round-bottomed flask (equipped with an N2 inlet, 60 mL pressure-equalizing dropping funnel, magnetic stirring bar, and rubber septum, and marked at approximately 190 mL) that has been pre-dried in an oven, under the protection of dry nitrogen gas.
2. 33.3 mL (270 mmol) of boron trifluoride ether compound was added, followed by cooling the mixture in a dry ice-acetone bath.
3. condense the DME into the DCM solution through a needle inserted through the rubber septum, which should remain just below the liquid level until the total volume of liquid reaches the 190mL mark.
4. add 24.1mL (307mmol) of epichlorohydrin dropwise over approximately 15 minutes with vigorous stirring (note: the mixture will become very viscous and may need to be rotated manually to ensure adequate stirring).
5. Remove the cooling bath and continue to vigorously stir the mixture overnight at room temperature.
6. Upon completion of the reaction, the resulting solid product was collected by filtration through a medium glass-filled Brinell's funnel, which had been pre oven-dried, under a stream of N2 gas.
7. The flask and filter were rinsed with dichloromethane (2 x 100mL) to remove the solids.
8. After drying under nitrogen protection, 29.4 g (98% yield) of free flowing white solid trimethoxonium tetrafluoroborate was obtained.
9. Store the product in a pre-oven dried glass vial in a nitrogen protected refrigerator.