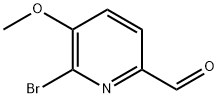
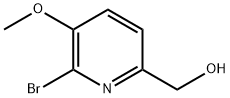
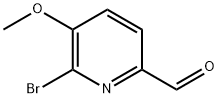
General procedure for the synthesis of 6-bromo-5-methoxypyridine-2-carboxaldehyde from (6-bromo-5-methoxypyridin-2-yl)methanol: (6-bromo-5-methoxypyridin-2-yl)methanol (2.30 g, 10 mmol) was dissolved in dichloromethane (50 mL) at room temperature, followed by addition of manganese dioxide (8.0 g). The reaction mixture was stirred continuously at room temperature for 24 hours. After completion of the reaction, the mixture was cooled to room temperature and the insoluble solids were removed by filtration. The filtrate was concentrated under reduced pressure to afford 6-bromo-5-methoxypyridine-2-carbaldehyde (1.35 g, 63% yield) as an off-white solid. Its nuclear magnetic resonance hydrogen spectrum (1H NMR, 400 MHz, CDCl3) data were as follows: δ 9.94 (s, 1H), 7.96 (d, J = 8.4 Hz, 1H), 7.27 (d, J = 8.0 Hz, 1H), 4.03 (s, 3H).