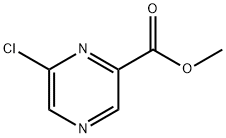
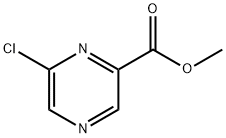
The product of Example 1D (7.18 g, 45.9 mmol) was taken as raw material and dissolved in dichlorosulfoxide (SOCl2, 50 mL, 687 mmol). The reaction mixture was heated to reflux and kept for 8 hours and then cooled to room temperature. Excess SOCl2 was evaporated under reduced pressure followed by slow addition of water (50 mL) at 0 °C to quench the reaction. The reaction mixture was neutralized with 1 M aqueous potassium carbonate (K2CO3, aq), followed by multiple extractions with dichloromethane (CH2Cl2, 5 × 100 mL). All organic phase extracts were combined, washed with saturated saline (100 mL), dried over anhydrous sodium sulfate (Na2SO4), filtered and concentrated under reduced pressure. The resulting residue was purified by silica gel column chromatography (eluent: 5% ethyl acetate in dichloromethane solution, Rf=0.35) to afford methyl 6-chloropyrazine-2-carboxylate as a thick oil, which gradually solidified after standing (7.16 g, 67% yield). The product was characterized by 1H NMR (DMSO-d6, 300 MHz): δ 3.94 (s, 3H), 9.07 (s, 1H), 9.19 (s, 1H). Mass spectrum (DCI/NH3) showed m/z 190 (M+NH4)+.