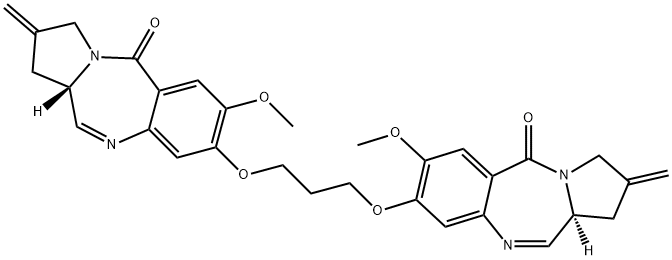
The general procedure for the synthesis of (11aS,11a'S)-8,8'-(propane-1,3-diylbis(oxy))bis(7-methoxy-2-methylene-2,3-dihydro-1H-benzo[e]pyrrolo[1,2-a][1,4]diazepin-5(11aH)-one) from the compound (CAS:1221969-15-8) was carried out as follows: the solid bis -SEM-dilactam 8 (100 mg, 0.12 mmol) was dissolved in a solvent mixture of ethanol (3 mL) and THF (3 mL). Lithium borohydride (22 mg, 2.3 mmol) was added all at once and the reaction mixture was stirred for 1 h under nitrogen protection. The completion of the reaction was monitored by LC/MS. The reaction mixture was partitioned between water (35 mL) and chloroform (50 mL). The organic phase was washed sequentially with water (35 mL), brine (35 mL), dried over magnesium sulfate and concentrated by rotary evaporation under reduced pressure. The residue was redissolved in a solvent mixture of chloroform (2 mL), ethanol (2 mL) and water (2.5 mL). Silica gel (4 g) was added and the mixture was stirred for 48 hours. The reaction mixture was filtered through a sintered funnel and washed with a solvent mixture of chloroform/methanol (90:10 v/v). The filtrate was extracted with chloroform, washed with brine, dried over magnesium sulfate and concentrated by rotary evaporation under reduced pressure. The residue was purified by fast chromatography (gradient elution: 100% CHCl3 to 97:3 v/v CHCl3/MeOH). The pure grades were collected and the solvent was removed by rotary evaporation under reduced pressure to give the target product SJG-136 as a mixture of imine and methanolamine methyl ether (46 mg, 70% yield). Analytical data: LC/MS 2.50 min (ES-) m/z (relative intensity) 555.06 ([M-H]-, 100); the analytical description is in agreement with the results previously published by Gregson et al. (J. Med. Chem. 2001, 44, 1161-1174). However, the observed [α]20D value was higher than the value reported in the literature: [α]20D = +766° (c = 0.37, HPLC CHCl3) (literature value [α]20D = +358° (c = 0.07, CHCl3)). This discrepancy is within expectations, as spin measurements are extremely sensitive to chloroform purity and the ratio of imine/carbamide adducts.
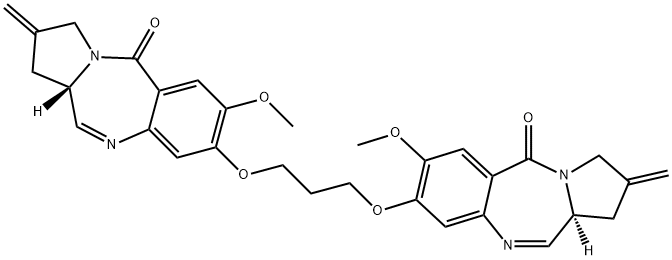

![1H-Pyrrolo[2,1-c][1,4]benzodiazepine-5,11(10H,11aH)-dione, 8,8'-[1,3-propanediylbis(oxy)]bis[2,3-dihydro-7-methoxy-2-methylene-10-[[2-(trimethylsilyl)ethoxy]methyl]-, (11aS,11'aS)-](/CAS/20210305/GIF/1221969-15-8.gif)