Definition
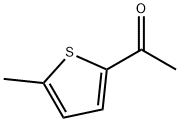
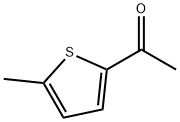
ChEBI: 2-acetyl-5-methylthiophene is a thiophene that is substituted by acetyl and methyl groups at positions 2 and 5, respectively. It has a role as a flavouring agent. It is an aromatic ketone, a member of thiophenes and a methyl ketone.
Synthesis
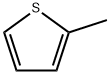
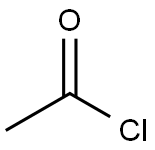
General procedure: 2-methylthiophene and acetyl chloride were used as raw materials for the synthesis of 2-acetyl-5-methylthiophene by Friedel-Crafts acylation reaction. The procedure was as follows: a double-necked round-bottomed flask equipped with a magnetic stirrer was charged with a pre-prepared MoO4 (AlCl22 (5 wt%, 10 mg)) catalyst, which was to be activated at 150 °C for 2 hours. Subsequently, 2-methylthiophene (1 g, 9.26 mmol) and acetyl chloride (5 equiv) were added to the reaction flask. The reaction mixture was stirred at room temperature until the reaction was completed and the reaction progress was monitored by thin layer chromatography (TLC). Upon completion of the reaction, the reaction mixture was filtered to isolate the solid catalyst, which could be recycled for the next Friedel-Crafts acylation reaction. The reaction mixture was washed sequentially with dichloromethane (DCM) and water and the process was repeated three times. The organic layer was collected, dried with anhydrous sodium sulfate and subsequently concentrated on a rotary evaporator to give the crude product. Finally, the crude product was purified by column chromatography to afford the target compound 2-acetyl-5-methylthiophene.
References
[1] European Journal of Organic Chemistry, 2010, # 31, p. 6033 - 6037
[2] European Journal of Organic Chemistry, 2016, vol. 2016, # 18, p. 3126 - 3129
[3] Journal of Nanoscience and Nanotechnology, 2015, vol. 15, # 10, p. 8243 - 8250
[4] Journal of Heterocyclic Chemistry, 1982, vol. 19, p. 701 - 702
[5] Chemische Berichte, 1886, vol. 19, p. 1858