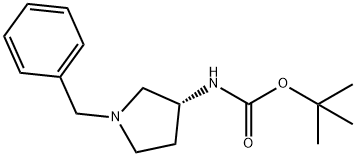
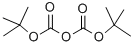
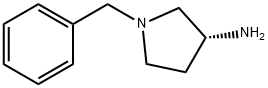
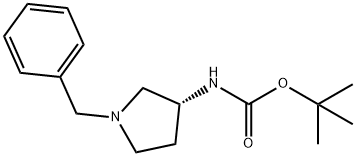
The general procedure for the synthesis of (R)-1-benzyl-3-(tert-butoxycarbonylamino)pyrrolidine from di-tert-butyl dicarbonate and (R)-1-benzyl-3-amino pyrrolidine was as follows: first, sodium bicarbonate (5.92 g, 70.5 mmol) was dissolved in 118 mL of deionized water, and then this solution was added to a 118 mL acetonitrile solution of (R)-(+)-benzylamino pyrrolidine 1a (5.00 g, 28.4 mmol) in 118 mL of acetonitrile solution. The mixture was stirred at room temperature for 10 minutes. Subsequently, di-tert-butyl dicarbonate (6.22 g, 28.5 mmol) was added and stirring was continued overnight at room temperature. After completion of the reaction, the reaction solution was concentrated under reduced pressure and the residue was extracted three times with dichloromethane. The organic layers were combined, dried over anhydrous sodium sulfate, filtered and concentrated. The residue was purified by fast column chromatography (eluent ratio methanol: dichloromethane = 2:98). Finally, the solvent was removed in vacuum to afford 4.24 g of tert-butyl (R)-(1-benzylpyrrolidin-3-yl)carbamate in 65.2% yield. The product was characterized by 1H NMR (400 MHz, CDCl3): δ 7.36-7.26 (m, 5H), 4.86 (bs, 1H), 4.18 (bs, 1H), 3.61 (s, 2H), 2.79 (bs, 1H), 2.65-2.61 (m, 1H), 2.54 (d, J=8.0 Hz, 1H), 2.34- 2.25 (m, 2H), 1.61-1.51 (m, 1H), 1.46 (s, 9H). The specific optical rotation [α]D was +2.5° (c 0.620, CHCl3).