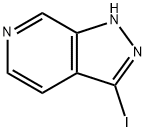
General procedure for the synthesis of 3-iodo-1H-pyrazolo[3,4-c]pyridine from 1H-pyrazolo[3,4-c]pyridine: 1H-pyrazolo[3,4-c]pyridine (300 mg, 2.52 mmol, 1.00 eq.), potassium hydroxide (KOH, 500 mg, 8.91 mmol, 3.50 eq.) and diiodomethane (1.28 g, 1.28 g, 2.00 eq.) were suspended in N,N-dimethylformamide (DMF, 10 mL). 5.04 mmol, 2.00 eq.) were suspended in N,N-dimethylformamide (DMF, 10 mL). The reaction mixture was stirred overnight at room temperature. After completion of the reaction, the reaction was quenched with water, the organic phase was extracted with ethyl acetate, the organic layer was dried with anhydrous sodium sulfate and concentrated under reduced pressure. The crude product was purified by silica gel column chromatography with the eluent ethyl acetate/petroleum ether (1:1, v/v) to afford 3-iodo-1H-pyrazolo[3,4-c]pyridine (577 mg, 84% yield) as a yellow solid.LC-MS (ESI, m/z): 246 [M + H]+.

![1H-PYRAZOLO[3,4-C]PYRIDINE](/CAS/GIF/271-47-6.gif)
![4-c]pyridine](/CAS/GIF/1082040-63-8.gif)