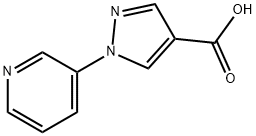
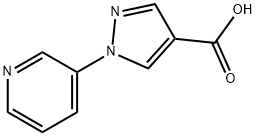
Stage 2: Synthesis of 1-(pyridin-3-yl)-1H-pyrazole-4-carboxylic acid
8.360 g (49.13 mmol) of 1-(pyridin-3-yl)-1H-pyrazole-4-carbonitrile was dissolved in 200 mL of dioxane and kept at a temperature of about 10 °C. Subsequently, 20 mL of water and 10.48 g of 45% sodium hydroxide aqueous solution were added. The reaction mixture was heated to reflux and maintained for 12 hours. Since the reaction was incomplete, 10.48 g of 45% aqueous sodium hydroxide solution was added additionally and reflux was continued for 12 hours. After completion of the reaction, the solution was cooled to room temperature and dioxane was removed by distillation under reduced pressure. The residue was diluted with a small amount of water and extracted with ethyl acetate. The aqueous phase was adjusted to pH 3 with concentrated hydrochloric acid at 0 °C. A precipitate was precipitated and collected by filtration to give 5.04 g of product. The filtrate was concentrated to half of the original volume and the precipitate was collected by filtration again to give another 2.29 g of product. The total yield was 7.33 g (72% of the theoretical yield).
Product characterization: logP(HCOOH) 0.50; 1H NMR ((CD3)2SO): δ 7.55 (m, 1H), 8.08 (s, 1H), 8.28 (m, 1H), 8.57 (m, 1H), 8.98 (s, 1H), 9.13 (m, 1H).