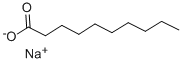
Description
Decanoic acid is a medium-chain saturated fatty acid. It is a non-competitive antagonist at AMPA receptors that selectively reduces glutamate-induced currents in
Xenopus oocytes expressing GluA2 and GluA3 subunit-containing AMPA receptors (IC
50 = 0.52 mM) over those expressing GluA1 (IC
50 = 2.09 mM) or GluA1 and GluA2 subunits (IC
50 = 1.16 mM). It inhibits epileptiform activity induced by pentylenetetrazole or low magnesium in rat hippocampal slices. Decanoic acid (1 mM) induces contractions in isolated guinea pig duodenum, an effect that can be blocked by the muscarinic acetylcholine receptor antagonist hyoscine, voltage-gated sodium channel inhibitor tetrodotoxin (Item Nos.
14964 |
14963), or M
2 muscarinic acetylcholine receptor antagonist hexamethonium . It increases the escape threshold in an orofacial mechanical stimulation test in rats when administered at a topical dose of 30% in ointment form, indicating analgesic activity. This effect can be blocked by the muscarinic acetylcholine receptor antagonist methoctramine . Plasma levels of decanoic acid are increased in patients with colorectal cancer when compared to patients with breast cancer or ulcerative colitis or without cancer.
Uses
Sodium Caprate is the sodium salt of capric acid. It functions as a binder, emulsifier, and anticaking agent.
Purification Methods
Neutralise sodium hydroxide by adding a slight excess of free decanoic acid and recovering the excess acid by Et2O extraction. The salt is recrystallised from the aqueous solution by adding pure acetone and repeating the steps several times, then drying the salt in an oven at ca 110o [Chaudhury & Awuwallia Trans Faraday Soc 77 3119 1981]. [Beilstein 2 IV 1041.]