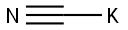
ЦИАНИД КАЛИЯ
- английское имяPOTASSIUM CYANIDE
- CAS №151-50-8
- CBNumberCB9387427
- ФормулаCKN
- мольный вес65.12
- EINECS205-792-3
- номер MDLMFCD00011397
- файл Mol151-50-8.mol
| Температура плавления | 634 °C (lit.) |
| Температура кипения | 1625 °C |
| плотность | 1.00 g/mL at 20 °C |
| Плотность накопления | 750kg/m3 |
| давление пара | 1.8hPa at 634.5℃ |
| Fp | 1625°C |
| температура хранения | Poison room |
| растворимость | H2O: 1 M at 20 °C, clear, colorless |
| пка | 9.36[at 20 ℃] |
| форма | Solid |
| цвет | White |
| Удельный вес | 1.52 |
| РН | 11-12 (20g/l, H2O, 20°C) |
| Растворимость в воде | Highly soluble in water. Soluble in methanol, glycerol, and formamide. Slightly soluble in ethanol. |
| Чувствительный | Hygroscopic |
| Мерк | 14,7626 |
| БРН | 4652394 |
| Пределы воздействия | TLV-TWA (measured as CN) skin 5 mg CN/m3 (ACGIH and OSHA); 5 mg CN/m3/ 10 min ceiling (NIOSH). |
| Стабильность | Stable. Incompatible with a variety of materials, including acids, iodine, peroxides, permanganates, alkaloids, chloral hydrate, metallic salts. Light and moisture sensitive. Contact with acid generates extremely toxic HCN gas. |
| LogP | -0.252 at 20℃ |
| Справочник по базе данных CAS | 151-50-8(CAS DataBase Reference) |
| Рейтинг продуктов питания EWG | 1 |
| FDA UNII | MQD255M2ZO |
| Система регистрации веществ EPA | Potassium cyanide (151-50-8) |
| UNSPSC Code | 12352302 |
| NACRES | NA.21 |
| Коды опасности | T+,N | |||||||||
| Заявления о рисках | 26/27/28-32-50/53 | |||||||||
| Заявления о безопасности | 7-28-29-45-60-61 | |||||||||
| РИДАДР | UN 1680 6.1/PG 1 | |||||||||
| OEL | Ceiling: 5 mg/m3 (4.7 ppm) [10-minute] [*Note: The REL also applies to other cyanides (as CN) except Hydrogen cyanide.] | |||||||||
| WGK Германия | 3 | |||||||||
| RTECS | TS8750000 | |||||||||
| F | 3-8-10-23 | |||||||||
| TSCA | Yes | |||||||||
| кода HS | 2837 19 00 | |||||||||
| Класс опасности | 6.1 | |||||||||
| Группа упаковки | I | |||||||||
| Банк данных об опасных веществах | 151-50-8(Hazardous Substances Data) | |||||||||
| Токсичность | LD50 orally in rats: 10 mg/kg (Hayes) | |||||||||
| ИДЛА | 25 mg/m3(as CN) | |||||||||
| NFPA 704: |
|
рисовальное письмо(GHS)
-
рисовальное письмо(GHS)




-
сигнальный язык
опасность
-
вредная бумага
H372:Поражает органы в результате многократного или продолжительного воздействия.
H300+H310+H330:Смертельно при проглатывании, при контакте с кожей или при вдыхании.
H410:Чрезвычайно токсично для водных организмов с долгосрочными последствиями.
H290:Может вызывать коррозию металлов.
-
оператор предупредительных мер
P262:Избегать попадания в глаза, на кожу или одежду.
P273:Избегать попадания в окружающую среду.
P280:Использовать перчатки/ средства защиты глаз/ лица.
P302+P352+P310:ПРИ ПОПАДАНИИ НА КОЖУ: Промыть большим количеством воды. Немедленно обратиться за медицинской помощью.
P304+P340+P310:ПРИ ВДЫХАНИИ: Свежий воздух, покой. Немедленно обратиться за медицинской помощью.
P314:В случае плохого самочувствия обратиться к врачу.
ЦИАНИД КАЛИЯ химические свойства, назначение, производство
Описание
Potassium cyanide, KCN, is a cyanide salt that is found as a white, amorphous, deliquescent lump or crystalline mass with a faint odor of bitter almonds. It is soluble in water and has a specific gravity of 1.52. It is a poison that is absorbed through the skin. Target organs are the same as for sodium cyanide. Reaction with acids releases flammable and toxic hydrogen cyanide gas. The four-digit UN identification number is 1680. The NFPA 704 designation is health 3, flammability 0, and reactivity 0. The primary uses are in gold and silver ore extraction, insecticides, fumigants, and electroplating.Химические свойства
Potassium cyanide are white lumps, granular powder, or colorless solution. It may be shipped as capsules, tablets, or pellets. Toxic hydrogen cyanide gas released by potassium cyanide has a distinctive, weak bitter almond odor, but many people cannot detect it; the odor does not provide adequate warning of hazardous concentrations.Использование
Potassium cyanide is a white granular salt made by the absorption of hydrogen cyanide in potassium hydroxide. It is soluble in both water and alcohol and a lethal poison. If mixed with acids it produces highly toxic hydrogen cyanide gas. This was the preferred fixing chemical for collodion positives because it contained no sulfides to darken the highlight silver. As a fixing agent cyanide was particularly effective. After dissolving the unexposed silver halides cyanide would also remove nonimage fog producing very clean shadow areas. Prolonged fixing would eventually remove image silver. Tincture of iodine was added to dilute solutions of potassium cyanide and used to remove unwanted non-image silver in photographic materials and to remove silver stains.Методы производства
Potassium cyanide was made by the Beilby process before the introduction of the neutralization or wet process. When made by the neutralization or wet process, it contains 99% KCN. Initially, potassium cyanide was used as a flux and later for electroplating, which was the single largest use in the 1990s. The demand for potassium cyanide was met by the ferrocyanide process until the latter part of the nineteenth century when the extraordinary demands of the gold mining industry for alkali cyanide resulted in the development of direct synthesis processes. When cheaper sodium cyanide became available, potassium cyanide was displaced in many uses.Общее описание
White amorphous lumps or a crystalline mass with a faint odor of bitter almonds. Density 1.52 g / cm3 Toxic by skin absorption through open wounds, by ingestion. Heating to decomposition produces toxic fumes. Used for gold and silver extraction, in chemical analysis, to make other chemicals, and as an insecticide.Реакции воздуха и воды
Deliquescent. Soluble in water. Dissolution releases some poisonous hydrogen cyanide gas. The amount is not hazardous except in an enclosed space. If the water is acidic, dangerous amounts of hydrogen cyanide form at once.Профиль реактивности
POTASSIUM CYANIDE is a basic salt and a reducing agent. Reacts with acids of all kinds to generate poisonous hydrogen cyanide gas. Can react violently with oxidizing agents: fusion with metal chlorates, perchlorates, nitrates, or nitrites can cause explosions [Bretherick 1979. p. 101]. A mixture with perchloryl fluoride may explode above 100°C. A mixture with nitrite salts may cause an explosion [Pieters 1957. p. 30]. Incompatible with iodine. Initiates the explosive decomposition of nitrogen trichloride.Опасность
A poison as absorbed by skin.Угроза здоровью
POTASSIUM CYANIDE is classified as super toxic. Probable oral lethal dose in humans is less than 5 mg/kg or less than a taste (7 drops) for a 150 lb. person. It is an eye and skin irritant. Poisonous in very small quantities; a taste is lethal.Пожароопасность
Contact with acid releases highly flammable hydrogen cyanide gas. Moisture may cause POTASSIUM CYANIDE to volatilize as hydrogen cyanide. When heated to decomposition, POTASSIUM CYANIDE emits very toxic fumes of cyanide and nitrogen oxides. Reacts with acids to produce hydrogen cyanide gas. Reacts with strong oxidizers such as nitrates and chlorates, nitrogen trichloride; perchloryl fluoride; sodium nitrate; acids; alkaloids; chloral hydrate; iodine. Avoid contact with acids.Промышленное использование
A white amorphous or crystalline solid of the composition KCN, potassium cyanide is employed for carbonizing steel for case hardening and for electroplating. For cyaniding steel the latter is immersed in a bath of molten cyanide and then quenched in water, or the cyanide is rubbed on the red-hot steel.Commercial potassium cyanide is likely to contain a proportion of sodium cyanide. Potassium ferrocyanide, or yellow prussiate of potash, can also be used for case-hardening steel. It has the compositionK4Fe(CN)6and comes in yellow crystals or powder. The nitrogen as well as the carbon enters the steel to form the hard case. Potassium ferricyanide, or red prussiate of potash, is a bright-red granular powder of the composition K3Fe(CN)6, used in photographic reducing solutions, in etching solutions, in blueprint paper, and in silvering mirrors. Redsol crystals is the name of this chemical for use as a reducer and mild oxidizing agent, or toner, for photography.
Профиль безопасности
A deadly human poison by ingestion. A experimental poison by ocular, subcutaneous, intravenous, intramuscular, and intraperitoneal routes. Experimental teratogenic and reproductive effects. Human systemic effects by ingestion: convulsions, pulse rate increase. Mutation data reported. Reacts with acids or acid fumes to liberate deadly HCN. When heated to decomposition it emits very toxic fumes of K2O , CN-, and NOx. See also CYANIDE.Возможный контакт
Used in electroplating, steel hardening; extraction of precious metals form ores; as a fumigant; in insecticides; a reagent in analytical chemistryПеревозки
UN1680 Potassium cyanide, Hazard Class: 6.1; Labels: 6.1-Poisonous materials.Методы очистки
A saturated solution in H2O-ethanol (1:3) at 60o is filtered and cooled to room temperature. Absolute EtOH is added, with stirring, until crystallisation ceases. The solution is again allowed to cool to room temperature (during 2-3hours), then the crystals are filtered off, washed with absolute EtOH, and dried, first at 70-80o for 2-3hours, then at 105o for 2hours [Brown et al. J Phys Chem 66 2426 1962]. It has also been purified by melting in a vacuum and by zone refining. HIGHLY POISONOUS.Несовместимости
A strong reducing agent; keep away from oxidizers. Potassium cyanide decomposes on contact with water, humidity, carbon dioxide, strong acids (such as hydrochloric, sulfuric, and nitric acids), and acid salts, producing highly toxic and highly flammable hydrogen cyanide gas. Potassium cyanide absorbs water from air (is hygroscopic or deliquescent); the aqueous solution is a strong base. Incompatible with organic anhydrides; isocyanates, alkylene oxides; epichlorohydrin, aldehydes, alcohols, glycols, phenols, cresols, caprolactum, strong oxidizers; nitrogen trichloride; sodium chlorate. Attacks aluminum, copper, zinc in the presence of moisture.Утилизация отходов
Consult with environmental regulatory agencies for guidance on acceptable disposal practices. Generators of waste containing this contaminant (≥100 kg/mo) must conform with EPA regulations governing storage, transportation, treatment, and waste disposal. In accordance with 40CFR165, follow recommendations for the disposal of pesticides and pesticide containers. Must be disposed properly by following package label directions or by contacting your local or federal environmental control agency, or by contacting your regional EPA office. Add strong alkaline hypochlorite and react for 24 hours. Then flush to sewer with large volumes of water.ЦИАНИД КАЛИЯ запасные части и сырье
сырьё
запасной предмет
- Изохинолин-1-карбонитрил
- 1-ЦИАНО-2-БЕНЗОИЛ-1,2-ДИГИДРОИЗОХИНОЛИН
- 2-METHYLBENZOYL CYANIDE
- 4- (N-ЦИАНОМЕТИЛ-N-НИТРОЗО) АМИНОМОРФОЛИН
- 2-NAPHTHALEN-1-YL-ETHYLAMINE
- 4-фтор-2-нитробензонитрила
- DL-бензоин
- Хинолин-2-карбонитрил
- 4-CHLORO-PYRIDINE-2-CARBONITRILE
- 2-AMINOPROPANENITRILE
- CYCLOLEUCINOL
- 1-метил-4-нитро-1H-имидазол-5-карбонитрил
- (S)-2-(1-BOC-2-пирролидинил)уксусная кислота
- DL-валин
- 3-морфолинозиднонимина гидрохлорид
- Сукцинодинитрил
- 2-(1,5-Dimethyl-1H-pyrazol-3-yl)acetonitrile ,97%
- 2-METHYLGLUTARIC ACID
- Серебро цианид
- Potassium dicyanoaurate
1of8