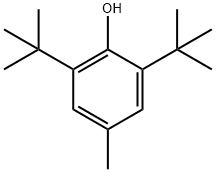
Бутилгидрокситолуол
- английское имяButylated Hydroxytoluene
- CAS №128-37-0
- CBNumberCB8355755
- ФормулаC15H24O
- мольный вес220.35
- EINECS204-881-4
- номер MDLMFCD00011644
- файл Mol128-37-0.mol
химическое свойство
| Температура плавления | 69-73 °C(lit.) |
| Температура кипения | 265 °C(lit.) |
| плотность | 1.048 |
| плотность пара | 7.6 (vs air) |
| давление пара | <0.01 mm Hg ( 20 °C) |
| показатель преломления | 1.4859 |
| FEMA | 2184 | BUTYLATED HYDROXYTOLUENE |
| Fp | 127 °C |
| температура хранения | 2-8°C |
| растворимость | methanol: 0.1 g/mL, clear, colorless |
| форма | Crystals |
| пка | pKa 14(H2O t = 25 c = 0.002 to 0.01) (Uncertain) |
| цвет | white |
| Запах | faint characteristic odor |
| Odor Type | phenolic |
| Растворимость в воде | insoluble |
| Мерк | 14,1548 |
| БРН | 1911640 |
| Пределы воздействия | ACGIH: TWA 2 mg/m3 NIOSH: TWA 10 mg/m3 |
| Стабильность | Stable, but light-sensitive. Incompatible with acid chlorides, acid anhydrides, brass, copper, copper alloys, steel, bases, oxidizing agents. Combustible. |
| ИнЧИКей | NLZUEZXRPGMBCV-UHFFFAOYSA-N |
| LogP | 5.2 |
| FDA 21 CFR | 172.115; 182.3173; 582.3173; 172.615; 175.105; 175.300; 177.1010; 181.24 |
| Вещества, добавляемые в пищу (ранее EAFUS) | BUTYLATED HYDROXYTOLUENE |
| Специальный комитет по веществам GRAS | Butylated Hydroxytoluene (BHT) |
| Справочник по базе данных CAS | 128-37-0(CAS DataBase Reference) |
| Рейтинг продуктов питания EWG | 4 |
| FDA UNII | 1P9D0Z171K |
| МАИР | 3 (Vol. 40, Sup 7) 1987 |
| Справочник по химии NIST | Butylated hydroxytoluene(128-37-0) |
| Система регистрации веществ EPA | 2,6-Di-tert-butyl-p-cresol (128-37-0) |
больше
| Коды опасности | Xn,N | |||||||||
| Заявления о рисках | 22-36/37/38-36/38-50/53 | |||||||||
| Заявления о безопасности | 26-36-37/39-61-60 | |||||||||
| РИДАДР | 3077 | |||||||||
| OEB | B | |||||||||
| OEL | TWA: 10 mg/m3 | |||||||||
| WGK Германия | 1 | |||||||||
| RTECS | GO7875000 | |||||||||
| F | 8-10-23 | |||||||||
| Температура самовоспламенения | 878 °F | |||||||||
| TSCA | Yes | |||||||||
| Класс опасности | 9 | |||||||||
| Группа упаковки | III | |||||||||
| кода HS | 29071900 | |||||||||
| Банк данных об опасных веществах | 128-37-0(Hazardous Substances Data) | |||||||||
| Токсичность | LD50 orally in mice: 1040 mg/kg (McOmie) | |||||||||
| NFPA 704: |
|
рисовальное письмо(GHS)
-
рисовальное письмо(GHS)

-
сигнальный язык
предупреждение
-
вредная бумага
H410:Чрезвычайно токсично для водных организмов с долгосрочными последствиями.
-
оператор предупредительных мер
P273:Избегать попадания в окружающую среду.
P391:Ликвидировать просыпания/проливы/утечки.
P501:Удалить содержимое/ контейнер на утвержденных станциях утилизации отходов.
Бутилгидрокситолуол химические свойства, назначение, производство
Описание
The antioxidant butylated hydroxytoluene is contained in food, adhesive glues, industrial oils and greases, including cutting fluids. Sensitization seems very rare.Химические свойства
Butylated hydroxytoluene occurs as a white or pale yellow crystalline solid or powder with a faint characteristic phenolic odor.Вхождение
Not reported found naturally.Использование
Butylated Hydroxytoluene (BHT) is an antioxidant that functions similarly to butylated hydroxyanisole (BHA) but is less stable at high temperatures. It is also termed 2,6-di-tert-butyl-para-cresol. See Butylated Hydroxyanisole.Определение
ChEBI: A member of the class of phenols that is 4-methylphenol substituted by tert-butyl groups at positions 2 and 6.Подготовка
Butylated Hydroxytoluene is produced commercially by the alkylation of para-cresol with isobutylene. Butylated Hydroxytoluene is also produced by several western European manufacturers, production/processing plants in Germany, France, the Netherlands, United Kingdom and Spain.Методы производства
Prepared by the reaction of p-cresol with isobutene.Общее описание
White crystalline solid.Реакции воздуха и воды
Insoluble in water.Профиль реактивности
Phenols, such as 2,6-Di-tert-butyl-4-methylphenol, do not behave as organic alcohols, as one might guess from the presence of a hydroxyl (-OH) group in their structure. Instead, they react as weak organic acids. Phenols and cresols are much weaker as acids than common carboxylic acids (phenol has Ka = 1.3 x 10^[-10]). These materials are incompatible with strong reducing substances such as hydrides, nitrides, alkali metals, and sulfides. Flammable gas (H2) is often generated, and the heat of the reaction may ignite the gas. Heat is also generated by the acid-base reaction between phenols and bases. Such heating may initiate polymerization of the organic compound. Phenols are sulfonated very readily (for example, by concentrated sulfuric acid at room temperature). The reactions generate heat. Phenols are also nitrated very rapidly, even by dilute nitric acid. Nitrated phenols often explode when heated. Many of them form metal salts that tend toward detonation by rather mild shock. May react with oxidizing materials.Угроза здоровью
2,6-Di-tert-butyl-p-cresol or Butylated Hydroxytoluene is of relatively low acute toxicity in animals, and there is no evidence of either acute or chronic effects among exposed workers.Пожароопасность
2,6-Di-tert-butyl-4-methylphenol is combustible.Фармацевтические приложения
Butylated hydroxytoluene is used as an antioxidant in cosmetics, foods, and pharmaceuticals. It is mainly used to delay or prevent the oxidative rancidity of fats and oils and to prevent loss of activity of oil-soluble vitamins.Butylated hydroxytoluene is also used at 0.5–1.0% w/w concentration in natural or synthetic rubber to provide enhanced color stability.
Butylated hydroxytoluene has some antiviral activity and has been used therapeutically to treat herpes simplex labialis.
Контактные аллергены
This antioxidant is contained in food, adhesive glues, industrial oils, and greases, including cutting fluids. Sensitization seems very rare.Канцерогенность
The IARC has determined that there is limited evidence for the carcinogenicity of Butylated Hydroxytoluene in experimental animals.Butylated Hydroxytoluene has given primarily negative results in a large number of in vivo and in vitro genotoxic assays.No significant reproductive effects were observed in three-generation toxicity studies in mice administered up to 0.4% in the diet.6 The 2003 ACGIH threshold limit valuetime- weighted average (TLV-TWA) for 2,6-ditert- butyl-p-cresol is 2mg/m3.Экологическая судьба
The metabolites of Butylated Hydroxytoluene can bind to cellular macromolecules, such as proteins and DNA, and cause toxicity.Возможный контакт
DBPC is used as a food and feed additive, flavor, and packaging material; as an antioxidant in human foods and animal feeds. It is also used as an antioxidant to sta- bilize petroleum fuels, rubber and vinyl plastics.Профиль безопасности
Poison by intraperitoneal andintravenous routes. Moderately toxic by ingestion. Anexperimental teratogen. Other experimental reproductiveeffects. A human skin irritant. A skin and eye irritant.Questionable carcinogen with experimental carcinogenicand.Безопасность
Butylated hydroxytoluene is readily absorbed from the gastrointestinal tract and is metabolized and excreted in the urine mainly as glucuronide conjugates of oxidation products. Although there have been some isolated reports of adverse skin reactions, butylated hydroxytoluene is generally regarded as nonirritant and nonsensitizing at the levels employed as an antioxidant.The WHO has set a temporary estimated acceptable daily intake for butylated hydroxytoluene at up to 125 μg/kg body-weight.
Ingestion of 4 g of butylated hydroxytoluene, although causing severe nausea and vomiting, has been reported to be nonfatal.
LD50 (guinea pig, oral): 10.7 g/kg
LD50 (mouse, IP): 0.14 g/kg
LD50 (mouse, IV): 0.18 g/kg
LD50 (mouse, oral): 0.65 g/kg
LD50 (rat, oral): 0.89 g/kg
хранилище
Exposure to light, moisture, and heat causes discoloration and a loss of activity. Butylated hydroxytoluene should be stored in a wellclosed container, protected from light, in a cool, dry place.Перевозки
UN2811 Toxic solids, organic, n.o.s., Hazard Class: 6.1; Labels: 6.1-Poisonous materials, Technical Name Required.Методы очистки
Dissolve Butylated Hydroxytoluene in n-hexane at room temperature, then cool with rapid stirring, to -60o. The precipitate is separated, redissolved in hexane, and the process is repeated until the mother liquor is no longer coloured. The final product is stored under N2 at 0o [Blanchard J Am Chem Soc 82 2014 1960]. It has also been recrystallised from EtOH, MeOH, *benzene, n-hexane, methylcyclohexane or pet ether (b 60-80o), and is dried in a vacuum. [Beilstein 6 IV 3511.]Несовместимости
Butylated hydroxytoluene is phenolic and undergoes reactions characteristic of phenols. It is incompatible with strong oxidizing agents such as peroxides and permanganates. Contact with oxidizing agents may cause spontaneous combustion. Iron salts cause discoloration with loss of activity. Heating with catalytic amounts of acids causes rapid decomposition with the release of the flammable gas isobutene.Регуляторный статус
GRAS listed. Accepted as a food additive in Europe. Included in the FDA Inactive Ingredients Database (IM and IV injections, nasal sprays, oral capsules and tablets, rectal, topical, and vaginal preparations). Included in nonparenteral medicines licensed in the UK. Included in the Canadian List of Acceptable Non-medicinal Ingredients.Бутилгидрокситолуол запасные части и сырье
Бутилгидрокситолуол поставщик
| поставщик | телефон | страна | номенклатура продукции | благоприятные условия | |
|---|---|---|---|---|---|
| +86-18066069912 +86-18066069912 |
China | 17 | 58 | ||
| +86-022-66880623 +8618622897568 |
China | 562 | 58 | ||
| 025-52279164 | China | 79 | 58 | ||
| +8619931165850 | China | 1000 | 58 | ||
| +86-371-86557731 +86-13613820652 |
China | 20314 | 58 | ||
| +86-029-81148696 +8615536356810 |
China | 1616 | 58 | ||
| +86 13288715578 +8613288715578 |
China | 12459 | 58 | ||
| +86-17531190177; +8617531190177 |
China | 6011 | 58 | ||
| +86-027-59207850 | China | 5984 | 58 | ||
| 0086 15632927689 +8615632927689 |
China | 984 | 58 |