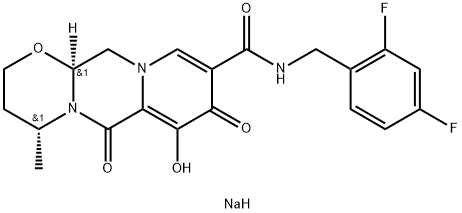
Dolutegravir sodium
- русский язык имя
- английское имяDolutegravir sodium
- CAS №1051375-19-9
- CBNumberCB52646573
- ФормулаC20H20F2N3NaO5
- мольный вес443.38
- EINECS812-620-6
- номер MDLMFCD28405599
- файл Mol1051375-19-9.mol
химическое свойство
| Температура плавления | >300oC |
| температура хранения | Hygroscopic, -20°C Freezer, Under inert atmosphere |
| растворимость | DMSO (Slightly, Heated), Methanol (Slightly, Heated) |
| форма | Solid |
| цвет | White to Green |
| Стабильность | Hygroscopic |
| ИнЧИКей | FWLDGCYHMZPGGI-SBBUJZKLNA-N |
| SMILES | O=C1N2[C@@H](CCO[C@@]2([H])CN2C=C(C(=O)NCC3C=CC(F)=CC=3F)C(=O)C(O)=C12)C.[NaH] |&1:3,7,r| |
| FDA UNII | 1Q1V9V5WYQ |
рисовальное письмо(GHS)
-
рисовальное письмо(GHS)

-
сигнальный язык
предупреждение
-
вредная бумага
H400:Чрезвычайно токсично для водных организмов.
H410:Чрезвычайно токсично для водных организмов с долгосрочными последствиями.
-
оператор предупредительных мер
P273:Избегать попадания в окружающую среду.
P391:Ликвидировать просыпания/проливы/утечки.
P501:Удалить содержимое/ контейнер на утвержденных станциях утилизации отходов.
Dolutegravir sodium химические свойства, назначение, производство
Описание
Dolutegravir, also known as DTG or dolutegravir sodium, is an antiretroviral therapy drug used to treat HIV infection. It belongs to the Integrase Strand Transfer inhibitor (INSTi) class of drugs and was fast-tracked by the FDA in February 2012. GlaxoSmithKline developed and markets dolutegravir sodium (Tivicay), which received FDA approval in August 2013 as a novel integrase inhibitor for HIV treatment, including adults undergoing their first treatment as well as those who have been treated with other integrase transfer strand inhibiting agents.Использование
Dolutegravir, a second-generation HIV-1 integrase strand transfer inhibitor, is commonly used along with other medications to manage HIV infection. Its potency in inhibiting HIV replication has been demonstrated in various cell types infected with a self-inactivating PHIV lentiviral vector, including peripheral blood mononuclear cells (PBMCs), MT-4 cells, and CIP4 cells.Определение
ChEBI: Dolutegravir sodium is an organic sodium salt that is the monosodium salt of dolutegravir. Used for treatment of HIV-1. It has a role as a HIV-1 integrase inhibitor. It contains a dolutegravir(1-).Побочные эффекты
Dolutegravir, an HIV medication, can lead to a variety of side effects. While some can be serious, many, such as nausea or sporadic dizziness, can be effectively managed. Dolutegravir may also cause alterations in your immune system, resulting in a condition known as immune reconstitution inflammatory syndrome (IRIS).clinicalinfo.hiv.gov/en/drugs/dolutegravir/patient
Синтез
The most likely process-scale synthesis of dolutegravir sodium, began with benzyl protection and alkylation of pyrone 46 with benzaldehyde, yielding alcohol 47 in 74% over 2 steps. Alcohol mesylation and in situ elimination provided the styrenyl olefin 48 in 94% yield, which further underwent an oxidative cleavage of the olefin to generate 49 by sequential addition of RuCl3/NaIO4 and NaClO2 (56% overall yield). Treatment of pyranone 49 with 3-amino-propane-2-diol (50) in ethanol at elevated temperatures delivered the corresponding pyridinone in 83% yield, and this was followed by esterification and sodium periodate-mediated diol cleavage to furnish intermediate 51 in 71% overall yield across the two-step sequence. l Next, the key ring-forming step in the synthesis of dolutegravir sodium consisted of cyclization of 51 with (R)-3- amino-butan-1-ol, a process which relies on substrate control to provide the desired tricyclic carbamoylpyridone system 52 in high stereoselectivity (20/1 in favor of the desired isomer).51 Previously, cyclization of systems such as 51 with unsubstituted amino alcohols were found to yield a mixture of diastereomeric products, therefore indicating the pivotal role of the chiral amino alcohol in influencing stereochemical bias during the overall cyclization step. In practice, reaction of 51 with (R)-3-amino-butan-1-ol at 90 ?? led to isolation of a single cyclization product 52, after recrystallization from EtOAc. From 52, N-bromosuccinimide (NBS) bromination and subsequent treatment with amine 53 under palladium-catalyzed amidocarbonylative conditions led to amide 54 in 75% yield over 2 steps. Finally, removal of the benzyl group and subsequent crystallization using sodium hydroxide in water and ethanol provided dolutegravir sodium (VII) in 99% yield.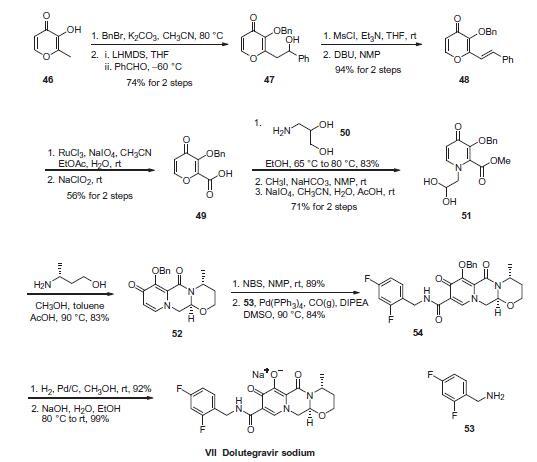
Dolutegravir sodium запасные части и сырье
Dolutegravir sodium поставщик
| поставщик | телефон | страна | номенклатура продукции | благоприятные условия | |
|---|---|---|---|---|---|
| +86-010-67886402 +8613611125266 |
China | 71 | 58 | ||
| +86-371-86557731 +86-13613820652 |
China | 20314 | 58 | ||
| 571-85586718 +8613336195806 |
China | 29798 | 60 | ||
| +86-371-66670886 | China | 16216 | 58 | ||
| +86-0371-55170693 +86-19937530512 |
China | 21670 | 55 | ||
| +86-0371-86658258 +8613203830695 |
China | 29897 | 58 | ||
| 18871490254 | CHINA | 28180 | 58 | ||
| +1-631-485-4226 | United States | 19553 | 58 | ||
| 0086-182-6772-3597 | CHINA | 419 | 58 | ||
| +86-023-6139-8061 +86-86-13650506873 |
China | 39916 | 58 |