Uses and Toxicity of Rifaximin
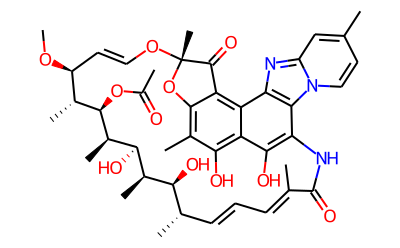
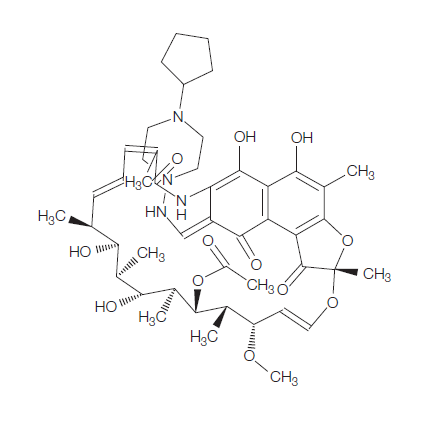
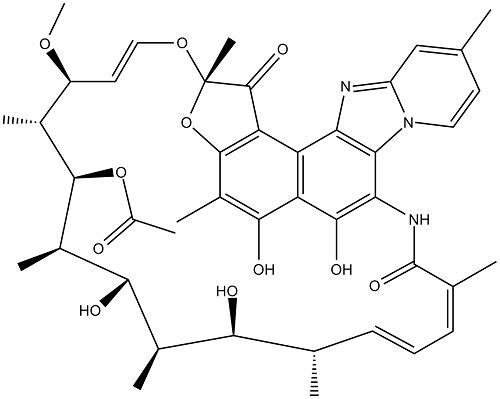
Rifaximin (4-deoxy-4u-methylpyrido[1u,2u-1,2]imidazole-[5,4c]-rifamycin SV) is a synthetic antimicrobial and derivative of rifamycin. It was first described, as compound L-105, in 1982 and marketed in Italy in 1987. Its empirical formula is C43H51N3O11 and its molecular weight is 785.9 Da; the chemical structure is shown in below.
Mechanism
Rifaximin acts by inhibiting bacterial RNA synthesis. It does so by binding the b unit of the bacterial DNA-dependent RNA polymerase and removing the catalytic magnesium ions from the RNA polymerase.
Uses
Rifaximin is a nonabsorbed drug with a high bioavailability in the gut, and fecal concentrations exceed MIC values for wide spectrum Grampositive and Gram-negative aerobic and anaerobic bacteria. These properties make it a potential drug for use in conditions involving the gastrointestinal tract, including infective diarrhea, inflammatory bowel disease, C. difficile infection, and hepatic encephalopathy. The lack of transcutaneous absorption has allowed it to be investigated for use in skin ailments and genital infections.
Bioavailability
Rifaximin is a nonabsorbable drug, with negligible absorption (less than 0.4%) from the gut. It is a molecule which is ionized at all pH values encountered in the gut, and as such is poorly absorbed. This property is as a result of an additional pyridoimidazole ring. Absorption of the drug is only minimally affected by gut inflammation In ulcerative colitis patients, the highest plasma level was 2–22.4 ng/ml. Absorption may be increased by high-fat foods, although this has not been found to be a significant effect.
When taken at 400 mg twice daily, rifaximin remains in stool at high concentrations over the next 5 days. Fecal concentrations reach as high as 4000–8000 mg/g after 3 days of administration. These concentrations are up to 250 times higher than the MIC90 for enteropathogens.
Toxicity
In animals, single-dose toxicity studies were performed in rats and mice by oral and intravenous administration. The LD50 in mouse is considered to be W2000 mg/kg, with one mouse dying at that dose (Scarpignato and Pelosini, 2005). In subacute toxicity studies, doses corresponding to 25 times the therapeutic dose in adults, 100 mg/kg, induced mild toxic effects, including acute gastroenteritis (Scarpignato and Pelosini, 2005). There was also a sex- and dose-dependent increase in total cholesterol in female animals, believed to be secondary to bile acid metabolism by altered gut flora (Scarpignato and Pelosini, 2005).
Related articles And Qustion
Lastest Price from Rifaximin manufacturers

US $0.00-0.00/kg2025-04-21
- CAS:
- 80621-81-4
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- 500 KGS

US $10.00/kg2025-04-21
- CAS:
- 80621-81-4
- Min. Order:
- 1kg
- Purity:
- 99.5%
- Supply Ability:
- 100 TON