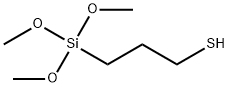
Trimethoxysilylpropanethiol for Material Modification
Trimethoxysilylpropanethiol is a commonly used sulfur-containing silane coupling agent. This molecule contains both hydrolyzable trimethoxysilyl groups and highly reactive thiol groups, which can chemically bond with hydroxyl groups on inorganic substrates and unsaturated bonds in rubber and resins, effectively enhancing the interfacial bonding strength between inorganic fillers and organic polymer materials. It is widely used in rubber reinforcement, composite bonding, metal surface corrosion protection, and coating modification. Trimethoxysilylpropanethiol can also serve as a crosslinking agent or surface modifier for hydrophobic modification and adhesion enhancement in glass and powder materials. This substance is slightly irritating and flammable; it must be stored in a sealed container, protected from moisture, and kept in a cool, well-ventilated area.

Fabrication of 1T-MoS2/Phenolic Photothermal Film
Buildings are estimated to contribute approximately 35% of global energy consumption, and a significant portion of carbon emissions originates from these structures, with an increase expected owing to population growth and urbanization. Unique opportunities are presented by molybdenum disulfide, a layered material. Within its layers, molybdenum atoms form covalent bonds with sulfur atoms, while interlayer connections are weak van der Waals forces, allowing MoS2 to be exfoliated into two-dimensional nanomaterials by various methods. In this work, a modified 1T-MoS2 (M1T-MoS2)/phenolic resin composite film was prepared. Due to the excellent dispersion of metallic-like M1T-MoS2 in the resin matrix, the film exhibits excellent broad-spectrum light shielding capabilities. 20 mL of 1T-MoS2 aqueous dispersion was added to 20 mL of deionized water and mixed with 40 mL of ethanol. Subsequently, 2 g of trimethoxysilylpropanethiol (TOPE) was introduced into the dispersion. After ultrasonication for 2 min, the mixture was stirred at 35 °C for 8 h. The resulting product was purified via filtration, then washed with ethanol. The obtained product was dispersed in ethanol and obtained a modified 1T-MoS2 (M1T-MoS2) dispersion with a mass fraction of approximately 4 wt %.As a result, trimethoxysilylpropanethiol molecules can be more easily adsorbed onto the MoS2 surface. And similar thiol-based surface modification strategies have also been widely employed in previous studies.[1]
The 1T-phase MoS2 nanosheets were synthesized by butyllithium intercalation and subsequently modified with TOPE in a water/ethanol mixed solvent (volume ratio of 1:1). The prepared M1T-MoS2 nanosheets were dispersed in ethanol to prevent aggregation and phase transformation. During the modification process, trimethoxysilylpropanethiol undergoes hydrolysis and polymerization, and the resulting products are subsequently anchored onto the surface of 1T-phase MoS2 nanosheets, driven by van der Waals interactions and the strong affinity of thiol groups for sulfur vacancies. The reflectance of MM-series films is lower than that of M-series films with the same filler content, and both series exhibit a decrease in reflectance as filler content increases. However, the change in reflectance is less pronounced compared to the variation in transmittance. This similarity in spectral shape indirectly indicates that trimethoxysilylpropanethiol modification does not significantly alter the intrinsic optical properties of MoS2. Since TOPE lacks a conjugated structure, it does not introduce absorption peaks in the visible and near-infrared regions. In this study, a modified 1T-phase molybdenum disulfide (M1T-MoS2)/phenolic resin composite film with broadband light-shielding capabilities was fabricated. Trimethoxysilylpropanethiol was used to modify the metallic-like1T-phase MoS2, enhancing its phase stability and dispersion in the resin. This study presents a method to stabilize 1T-phase MoS2 nanosheets in composite materials, offering potential for expanding their application in energy, composites, and wearable devices.
C18-Sulfonic Group Dual Modification Chromatographic Stationary Phase
A C18-sulfonic group dual modification chromatographic stationary phase was prepared by one-pot reaction to modify the surface of silica with two modifier of octadecyltrichlorosilane (OTS) and trimethoxysilylpropanethiol (MPS), and then oxidize the thiol group. Under the optimized reaction conditions, the stationary phase with a mole ratio of 3:7 between C18 and sulfonic group was obtained. The morphology and feature of the stationary phase were characterized by scanning electronic microscopy, elemental analysis and infrared spectra. The chromatographic properties of the prepared stationary phase were systematically investigated in different separation modes. Five alkyl benzenes and three nucleosides were separated successfully by reversed phase chromatography and hydrophilic interaction chromatography, respectively.[2]
The bonding rates of octadecyltrichlorosilane and trimethoxysilylpropanethiol were investigated at different mole ratios between octadecyltrichlorosilane and trimethoxysilylpropanethiol that were of 1:1, 2:1, 4:1. Finally, the mole ratio of 1:1 was the best choice for the following experiments and the specific steps were as follows. Typically, 3 g of silica, 50 mL of hydrochloric acid and 20 mL of deionized water were mixed and refluxed with continuous stirring for 8 h at 110 °C. Then the reaction solution was filtrated and washed 5 times with 500 mL of deionized water. The activated silica (AS) was gotten after dried at 120 ºC in vacuum overnight. Subsequently, the AS was dropped in 50 mL of toluene, 1.0 mL of OTS, 0.5 mL of trimethoxysilylpropanethiol and 50 μL of trimethylamine and refluxed with continuous stirring for 24 h at 100 ºC under nitrogen protection. Then the reaction solution was filtrated and washed successively with 100 mL of dichloromethane, 50 mL of methanol, 100 mL of deionized water and 50 mL of methanol before drying at 80 ºC in vacuum overnight to achieve C18-mercapto silica (octadecyl-mercapto silica, OMS). Subsequently, 15 mL of methanol, 50 mL of hydrogen peroxide and 20 μL of sulfuric acid were added in OMS. The mixed solution was then stirred for 20 h at 25 ºC and filtrated. Finally, the products, called C18-sulfonic silica (octadecyl- sulfonic silica, OSS), were washed with 100 mL of deionized water and 100 mL of methanol before drying at 80 ºC overnight.
References
[1]Qing Zeng, & Bo You*, (2025). Fabrication of 1T-MoS2/Phenolic Photothermal Film for Broad-Spectrum Light-Shading Application with Low Reflectivity and High Imaging Clarity. ACS Applied Materials & Interfaces, 17 35, 49884–49896. https://doi.org/10.1021/acsami.5c11345
[2]Tao TANG . (2017). Preparation and Evaluation of a C18-Sulfonic Group Dual Modification Chromatographic Stationary Phase. Chinese Journal of Analytical Chemistry, 45 1, Pages 56-60. https://doi.org/10.1016/S1872-2040(16)60989-1
Lastest Price from Trimethoxysilylpropanethiol manufacturers

US $0.00-0.00/kg2025-06-17
- CAS:
- 4420-74-0
- Min. Order:
- 0.01kg
- Purity:
- 99.99%
- Supply Ability:
- 200kg

US $10.00/KG2025-04-21
- CAS:
- 4420-74-0
- Min. Order:
- 1KG
- Purity:
- 99%
- Supply Ability:
- 100 mt