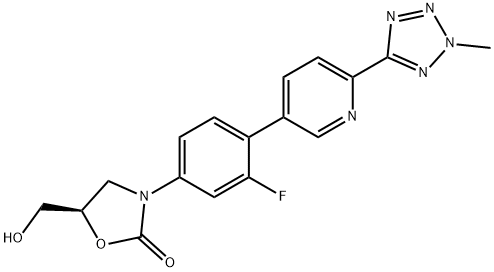
Synthesis of Tedizolid
Tedizolid is the first FDA-approved second-generation oxazolidinone antibiotic, which is clinically used to treat skin and skin tissue infections, and its efficacy is non-inferior to linezolid. Compared with linezolid, the safety of dezolid has been improved to a certain extent, and the incidence of gastrointestinal adverse reactions is lower than that of linezolid. The in vitro inhibitory activity of tedizolid on some bacteria is 2 to 8 times higher than that of linezolid, the treatment period is shortened by 40%, and the incidence of drug resistance is lower than that of linezolid.
Antimicrobial spectrum
Tedizolid is effective against a variety of gram-positive bacteria in vitro, including: streptococci; enterococci, including vancomycin-resistant enterococci (VRE); coagulase-negative staphylococci; methicillin-sensitive gold Staphylococcus aureus; methicillin-resistant S. aureus (MRSA); Bacillus; Corynebacterium; Listeria monocytogenes ).
Advantage
Tedizolid, a second-generation oxazolidinone antibacterial drug, was approved for acute skin and soft tissue infections based on two clinical trials that demonstrated its efficacy was non-inferior to linezolid. Tedizolid was more well tolerated than linezolid, and a meta-analysis showed that tedizolid had a lower incidence of thrombocytopenia.
Synthesis method
A kind of purification method of high-purity tedizolid phosphate, it comprises the steps:
(1) with dimethyl sulfoxide heating and dissolving the tedezolid phosphate crude product, after dissolving and clarifying, add polar protic solvent wherein, control temperature and stir, cooling carries out crystallization, suction filtration;
(2) adding the filter cake obtained in step (1) into water, adjusting the pH of the solution to alkalinity with an aqueous solution of sodium hydroxide or potassium hydroxide, then slowly adding it to the water-soluble aprotic organic solvent for crystallization, suction filtration, and After crystallization in a water-soluble aprotic organic solvent and suction filtration, the obtained filter cake is added to a mixed solvent of water and tetrahydrofuran, the pH is quickly adjusted to 1-2 with aqueous hydrochloric acid, stirred for crystallization, and suction filtered;
(3) after the last gained filter cake of step (2) is dissolved in dimethyl sulfoxide, it is slowly added to the mixed solvent of water and ethanol of temperature control, cooling and crystallization, suction filtration, and drying to obtain high-purity tedizolid phosphate;
Wherein, the polar protic solvent described in step (1) is a mixed solvent of water, water and methanol, or a mixed solvent of water and ethanol;
The control temperature described in step (1) means that the control temperature is 50°C to 80°C;
The described stirring time of step (1) is 3h~15h;
The step (2) of adjusting the pH of the solution to alkaline refers to adjusting the pH of the solution to be 7-9;
The water-soluble aprotic organic solvent described in step (2) is selected from the one in tetrahydrofuran, acetonitrile or acetone;
The volume ratio of the water-soluble aprotic organic solvent described in step (2) to water is 5:1 to 10:1;
The temperature-controlled water and ethanol described in step (3) refer to water and ethanol whose controlled temperature is 40-70 °C;
The temperature of the crystallization described in step (3) is 10-50°C.
See also
Lastest Price from Torezolid manufacturers

US $6.00/kg2025-04-21
- CAS:
- 856866-72-3
- Min. Order:
- 1kg
- Purity:
- 0.99
- Supply Ability:
- 1000

US $10.00/KG2025-04-21
- CAS:
- 856866-72-3
- Min. Order:
- 1KG
- Purity:
- 99%
- Supply Ability:
- 10 mt