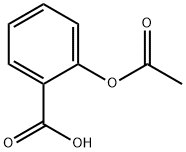
Synthesis of Aspirin
Aspirin is an effective analgesic (pain reliever), antipyretic (fever reducer) and anti-inflammatory agent and is one of the most widely used non-prescription drugs. The use of aspirin had its origin in the 18th century, when it was found that an extract from the bark of willow trees was useful in reducing pain and fever. The active ingredient in willow bark was later found to be salicylic acid. The structure of salicylic acid is shown below. Although salicylic acid was effective at reducing pain and fever, it also had some unpleasant side effects. It is irritating to the lining of the mouth, esophagus, and stomach, and can cause hemorrhaging of the stomach lining. In 1899, the Bayer Company in Germany patented a drug they called aspirin, which was a modification of salicylic acid.
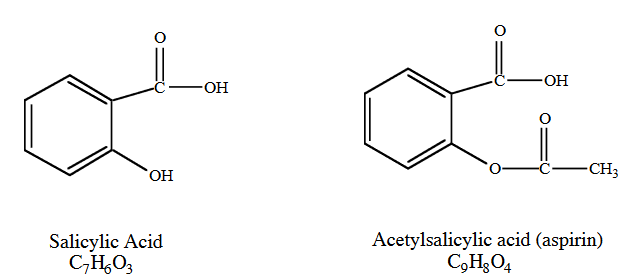
Salicylic acid contains a phenol group, and phenols are known to be irritating. The Bayer Company replaced the phenol group with an ester group. This esterified compound (acetylsalicylic acid, also known as aspirin) was shown to be much less irritating than salicylic acid. Unfortunately, it is still irritating to the stomach and can cause hemorrhaging of the stomach walls.
An aspirin tablet contains a small amount of aspirin (usually 300-400 mg) in a starch “binder” and sometimes contains other ingredients like caffeine and buffers. When aspirin is ingested, it is broken down to salicylic acid by the basic conditions in the small intestine. It is then absorbed into the bloodstream.
Aspirin can be made by reacting salicylic acid with acetic acid in the presence of an acid catalyst. The phenol group on the salicylic acid forms an ester with the carboxyl group on the acetic acid. However, this reaction is slow and has a relatively low yield. If acetic anhydride is used instead of acetic acid, the reaction is much faster and has a higher yield (since acetic anhydride is much more reactive than acetic acid).
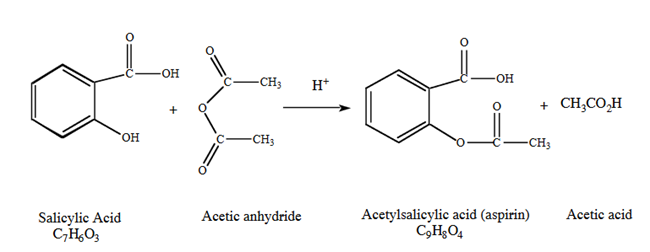
In this experiment, the salicylic acid is the limiting reactant and the acetic anhydride is in excess. After the reaction heating period is over, the excess unreacted acetic anhydride will be destroyed by the addition of water to the mixture: water reacts with acetic anhydride to form 2 molecules of acetic acid, according to the reaction shown below.
When the esterification reaction is complete, water will be added to the mixture. This will cause the precipitation of the acetylsalicylic acid and will react with any remaining acetic anhydride. The solid aspirin will be collected using vacuum filtration. Any other reaction ingredients that are soluble (this includes acetic acid, phosphoric acid, and water) will pass through the filter paper.
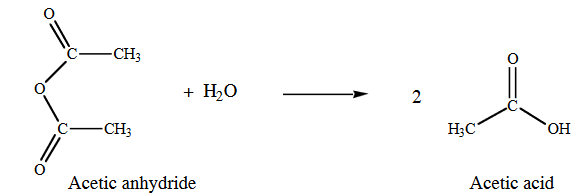
The collected aspirin will be tested for its purity using FeCl3 (aq). Iron (III) ion reacts with phenols to form a purple complex. Salicylic acid contains a phenol group, but acetylsalicylic acid does not. Therefore, if you add FeCl3 to an aspirin sample and you see a purple color, it means that there is still some salicylic acid present and the sample is impure.The aspirin collected will then be purified by recrystallization. In this purification method, the crude aspirin will be dissolved in a small amount of warm ethanol. Water will then be added and the solution will be cooled slowly and then chilled. The acetylsalicylic acid will recrystallize, and the solid impurities (unreacted salicylic acid) should remain dissolved in the solution. The solid aspirin will again be collected using vacuum filtration and tested for purity. This aspirin should be more pure than the original aspirin.The final product will be dried and weighed and the theoretical and percent yields will be calculated.
Related articles And Qustion
See also
Lastest Price from Acetylsalicylic acid manufacturers

US $0.00-0.00/kg2025-07-23
- CAS:
- 50-78-2
- Min. Order:
- 1kg
- Purity:
- 99.5%-101%
- Supply Ability:
- 500KG

US $1.00/KG2025-04-21
- CAS:
- 50-78-2
- Min. Order:
- 1KG
- Purity:
- 99%
- Supply Ability:
- 10 mt