Refractories: Classification and Properties
Classification of Refractories
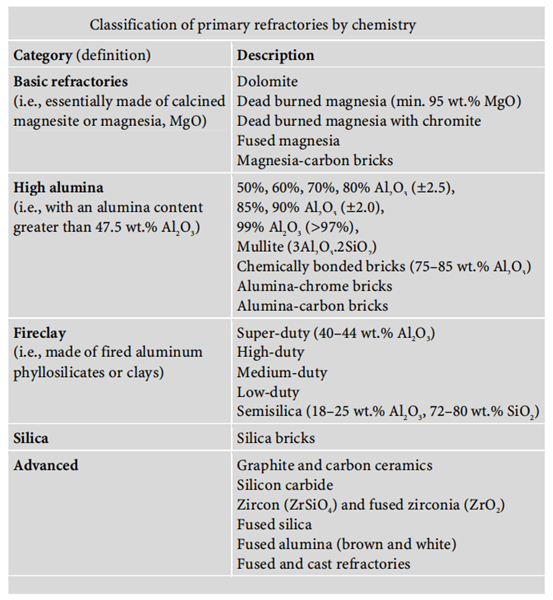
The classification of refractories can be approached in a number of different ways: chemical composition, type of applications, or operating temperature range. Classification of primary refractories by chemistry:
Properties of Refractories
Silica brick
The earliest silica bricks were composed of crushed minerals of 90 wt.% silica, with as much as 3.5 wt.% flux materials (i.e., usually CaO), and fired at about 1010°C. They can serve in highertemperature service to about 1093°C and are more resistant to thermal shock due to their greater porosity, as high as 16%. When used in chemical service, those of the highest silica content (not below 98 wt.% SiO2) should be used. The purity of the silica and its percentage of alkali, along with the manufacturing techniques, determine the uniformity or the wideness of ranges of the physical properties. Ranges of chemical composition of silica brick are 98.6 to 99.6 wt.% SiO2, 0.2 to 0.5 wt.% Al2O3, 0.02 to 0.3 wt.% Fe2O3, 0.02 to 0.1 wt.% MgO, 0.02 to 0.03 wt.%CaO, and 0.01 to 0.2 wt.% (Na2O, K2O, Li2O).
Silica brick serves well and for long periods in acid service, except for hydrofluoric, without noticeable damage, showing greater resistance, especially to strong hot mineral acids, and particularly phosphoric rather than acid brick, and in halogen exposures, except fluorine, solvents, and organic chemical exposures. Silica bricks are not recommended for service in strong alkali environments. They also exhibit better shock resistance than shale or fireclay acid brick, but they have lower strength and abrasion resistance.
Porcelain brick
Porcelain bricks are made from high-fired clays, the temperature of firing depending on the amount of alumina in the clay, 15% to 38% usually at ca. 1200 to 1300°C, 85% at 1500 to 1550°C, and 95 to 98% at 1600 to 1700°C. The bodies of these bricks are extremely dense and nonporous, with zero absorption, and a Mohs hardness ranging from 6 to 9 for 99 wt.% alumina. As alumina content increases, Mohs hardness, the maximum service temperature, and chemical resistance increase. Major uses of porcelain include: the lining of ball mills, where they will outlast almost all other abrasion-resistant linings, and employment (glazed) as pole line hardware for the power industry where, exposed to abrasion, weathering, and cycling temperature changes, they outlast all other materials in similar service. Due to the high cost of porcelain brick, they are used sparingly in the process industries, chiefly in dye manufacture, due to their density for the prevention of interbatch contamination and ease of cleaning. The use of porcelain brick is primarily limited by its cost.
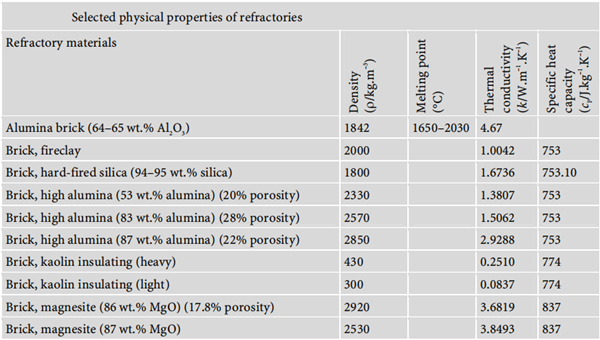
Selected physical properties of refractories: