Properties and Reactions of Pyrrole
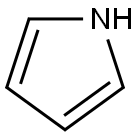
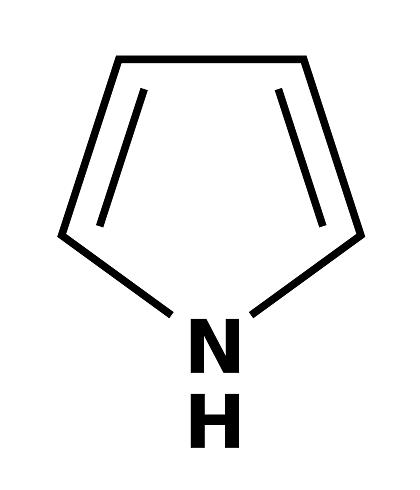
Pyrrole is a heterocyclic aromatic organic compound, a five-membered ring with the formula C4H4NH.It is a colorless volatile liquid that darkens readily upon exposure to air. Substituted derivatives are also called pyrroles, e.g., N-methylpyrrole, C4H4NCH3. Porphobilinogen, a trisubstituted pyrrole, is the biosynthetic precursor to many natural products such as heme.
Properties
Pyrrole has a nutty odor. Pyrrole is a 5-membered aromatic heterocycle, like furan and thiophene. Unlike furan and thiophene, it has a dipole in which the positive end lies on the side of the heteroatom, with a dipole moment of 1.58 D. In CDCl3, it has chemical shifts at 6.68 (H2, H5) and 6.22 (H3, H4). Pyrrole is weakly basic, with a conjugate acid pKa of −3.8. The most thermodynamically stable pyrrolium cation (C4H6N+) is formed by protonation at the 2 position.
Pyrrole is very much less basic than secondary amines but much more acidic. Pyrrole is, however, still a very weak acid (pKa = 17.5). The nitrogen-bound proton can be abstracted from pyrrole by the use of strong bases such as sodium amide in liquid ammonia and n-butyllithium in hexane. Reaction of pyrrole with Grignard reagents results in the formation of halomagnesyl derivatives 170. These nucleophiles have ambident properties and can react with electrophiles at nitrogen, C(2) or C(3). N-Unsubstituted indoles similarly form metal derivatives when treated with a variety of reagents such as n-butyllithium, sodamide, Grignard reagents and thallium(I) ethoxide
Reactions
Pyrrole, being a π-rich heterocycle, is not easily reduced catalytically. However, catalytic reduction is accomplished at moderate temperature and pressure using Pd, Pt, or Raney Ni catalysts. The presence of an electron-withdrawing N-substituent facilitates reduction of the pyrrole ring, while in the case of C-substituted pyrrole, preferential autolytic reduction of substituent takes place.
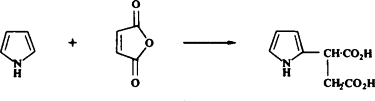
Catalysts such as copper chromite are capable of reducing the pyrrole ring as well as substituents present on the ring. However, catalysts such as rhodium and aluminum only reduce the pyrrole ring leaving substituents unaffected.
Chemical reduction of pyrrole with Zn/HCl gave a mixture of ∆1-, ∆2-, and ∆3-pyrrolines in which the latter was the major product. ∆1- and ∆2-Pyrrolines further reduced to pyrrolidine. Reduction of pyrrole with hydriodic acid and phosphorus gave pyrrolidine. However, when Na–ethanol was used as reducing agent, substituents were reduced preferentially compared to the pyrrole ring. Sodium-ethanol reduction of 1-arylpyrrole afforded 1-(1-pyrrollyl)cyclohexa-1,4-diene by reducing the aryl ring leaving the pyrrole ring unaffected.
You may like
Related articles And Qustion
See also
Lastest Price from Pyrrole manufacturers

US $5.00-2.00/KG2024-10-11
- CAS:
- 109-97-7
- Min. Order:
- 1KG
- Purity:
- 99%
- Supply Ability:
- 10000kg
US $30.00-28.00/Kg2023-05-31
- CAS:
- 109-97-7
- Min. Order:
- 1Kg
- Purity:
- 99.0% up
- Supply Ability:
- 50 tons per month