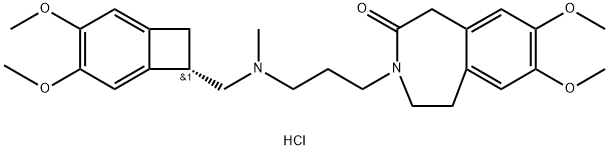
Ivabradine Hydrochloride in Angina and Cardiac Therapy
Research background
Treatment for angina includes reducing heart rate with agents such as β-adrenergic blockers and calcium channel blockers. However, these agents also exert other unwanted activities such as negative inotropic and hypotensive effects which could have serious consequences. Thus, the search for novel heart rate-reducing compounds without unwanted inotropic effects was initiated. A new class of specific bradycardic agents act specifically on the SA node by directly interacting with the pacemaking cell of the SA node and the hyperpolarization-activated If, the primary pacemaking current of the SA node. Ivabradine hydrochloride is one such bradycardic agent to emerge that has been shown to specifically and selectively interact with f-channels of pacemaker cells to block If and consequently reduce the speed of diastolic depolarization and decrease heart rate. Due to its potent preclinical action, ivabradine hydrochloride was selected for further development as a treatment for stable angina and underlying myocardial ischemia as well as supraventricular arrhythmias. [1]
Mechanism
The sinoatrial node is unique in that its cells have an innate ability to generate a cyclical change in their resting membrane potential, which drives it toward the threshold needed for spontaneous depolarization. This depolarization, in turn, generates repetitive, spontaneous action potentials accounting for its automaticity. This depolarization is initiated by the opening of specific ion channels that conduct a slow, inward-depolarizing mixed sodium-potassium current, referred to as the pacemaker or “funny” current. If is generated via a nonselective, hyperpolarization-activated cyclic nucleotide–gated transmembrane channel (Central Illustration). Ivabradine hydrochloride blocks the intracellular aspect of this transmembrane channel by inhibiting cation movement with a high degree of selectivity, leading to a reduction in the slope of the diastolic depolarization of the pacemaker action potential, thereby slowing the heart rate. Ivabradine hydrochloride blocks the channel in its open state, creating a particularly favorable attribute, use dependence (i.e., it becomes more potent at faster heart rates). Ivabradine hydrochloride causes a dose-dependent reduction in heart rate and, as a consequence of its specific mechanism of action, it is able to do so without affecting cardiac inotropy or systemic vascular resistance. [2]
Preparation and Characterization of Ivabradine HCl Transfersomes for Enhanced Transdermal Delivery
Ivabradine hydrochloride (IVB) is used in treatment of stable angina pectoris. The aim of the work was to prepare a once daily transdermal film of transfersomes loaded IVB. Transfersomes were prepared by modified ethanol injection method using three formulation factors namely: surfactant type, drug: phospholipid ratio and surfactant: phospholipid ratio. The transfersomes were characterized for entrapment efficiency, particle size, zeta potential, in vitro drug release, optical and transmission electron microscopy imaging, differential scanning calorimetry (DSC) and Fourier transform infrared spectroscopy (FTIR). The best transfersomes showed optimal properties as well as pure drug were further formulated in transdermal film and characterized for content uniformity, folding endurance, weight and thickness, moisture content, moisture uptake, tensile strength and permeation through excised ear rabbit skin. Pharmacokinetic parameters of transfersomes incorporated film were compared with oral solution of the same drug dose. A maximum of about 82.03% entrapment efficiency, size of 206.7 nm, zeta potential of -88.3 Mv and 66.7% of drug release after 24 h were obtained from sodium lauryl sulphate based transfersomes at drug: phospholipid and surfactant: phospholipid ratio of 1:20 and 75:25 w/w, respectively. Films contained IVB transfersomes exhibited improved permeability and skin retention when compared with pure drug as well as enhanced pharmacokinetic parameters.[3]
IVB-loaded transfersomes were prepared successfully by using ethanol injection technique. Selection of surfactant, drug: phospholipid ratio and surfactant: phospholipid ratio are critical factors to obtain the desired formulation. Formulation T8 which based on sodium lauryl sulphate at drug: phospholipid ratio of 1:20 and surfactant: phospho-lipid ratio of 75:25 was found to have best sustained release behavior at pH 7.4 with desirable properties in terms of entrapment efficiency (82.03 ± 0.61%), particle size (206.7 nm) and zeta potential (-88.3 ± 8.9mV). [3]

References:
[1] Sorbera, L. A., & Castaner, J. (2003). Ivabradine hydrochloride. Drugs of the Future, 28(7).
[2] Koruth, J. S., Lala, A., Pinney, S., Reddy, V. Y., & Dukkipati, S. R. (2017). The clinical use of ivabradine. Journal of the American College of Cardiology, 70(14), 1777-1784.
[3] Balata, G. F., Faisal, M. M., Elghamry, H. A., & Sabry, S. A. (2020). Preparation and characterization of ivabradine HCl transfersomes for enhanced transdermal delivery. Journal of drug delivery science and technology, 60, 101921.
You may like
See also
Lastest Price from Ivabradine hydrochloride manufacturers

US $1.00/kg2025-07-09
- CAS:
- 148849-67-6
- Min. Order:
- 1kg
- Purity:
- 99%
- Supply Ability:
- 1

US $5.00-0.50/KG2025-06-05
- CAS:
- 148849-67-6
- Min. Order:
- 1KG
- Purity:
- 99% hplc
- Supply Ability:
- 500TONS