Introduction of Ritipenem
DESCRIPTION
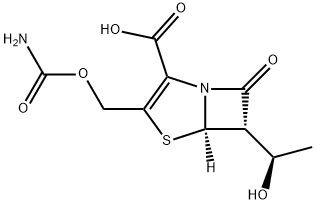
Ritipenem is a synthetic b-lactam antibiotic of the penem class that has a broad antimicrobial spectrum and high resistance to b-lactamases. Since it is stable in relation to renal dehydropeptidase-I, ritipenem does not require concomitant administration of an inhibitor of this enzyme.
Ritipenem acoxil (FCE 22891) is the acetoxymethyl ester prodrug which is orally available, but is hydrolyzed very rapidly to the active component ritipenem (FCE 22101), as well as some other metabolites. Less than 50% of ritipenem acoxil is absorbed in the intestinal tract, and this fraction is completely hydrolyzed to ritipenem during transport across the gastrointestinal mucosa.
Ritipenem acoxil does not have antimicrobial activity unless hydrolyzed to ritipenem. Ritipenem is notable as one of the early orally available carbapenems – it is manufactured by Tanabe Seiyaku in the ritipenem acoxil prodrug form (500 mg ritipenem acoxil is equivalent to about 399.75 mg ritipenem anhydrous acid). As of 2010, it is not FDA approved in the USA.
MECHANISM OF DRUG ACTION
In common with other b-lactams, ritipenem interacts with the penicillin-binding proteins (PBPs). In E. coli, the main target of ritipenem is PBP2, and PBP3 is also equivalent to PBP2 in the Enterobacteriaceae. In vitro studies suggest that ritipenem has affinity for the PBP2 of methicillin-susceptible S. aureus and PBP2u of methicillin-resistant S. aureus.
PHARMACOKINETICS AND PHARMACODYNAMICS
Oral ritipenem (ritipenem acoxil) is only about 30–40% bioavailable, producing a mean AUC of 497 mg/l min in healthy volunteers. However, some intersubject variability has been noted – possibly due to either presystemic metabolism of ritipenem and/or transformation of the prodrug to compounds other than ritipenem.
Animal studies revealed that the major route of ritipenem elimination was via the urine. In a rat study, 90.2% of ritipenem and its metabolites were excreted in the urine and 4.5% were excreted in the feces after 72 hours of administration (Battaglia et al., 1992). In the same study, when a 1:1 combination with ritipenem and cilastatin was administered to rats, the ratio of ritipenem in the urine during 0–8 hours was increased from 35.3% to 49.4%.
In a human study with intravenous co-administration of ritipenem with imipenem–cilastatin, the urinary recovery of ritipenem (80.7%) was significantly higher than with ritipenem alone (36.9%) (Norrby et al., 1990). When 4 mg/kg of ritipenem was infused intravenously in healthy volunteers over 10 minutes, the urinary recovery was 26.6% and 0.9% of the dose at the 0- to 2-hour and 2- to 4-hour collection intervals, respectively. No ritipenem was detected in the urine after a 4- to 6-hour infusion. The major metabolite found in urine accounted for 42.4% of the dose. Efthymiopoulos et al. (1992) reported that, in a study of four healthy male volunteers administered 500 mg of oral ritipenem acoxil, the average amount of ritipenem excreted in urine and feces corresponded to 9% and 1.6% of the dose, respectively.
TOXICITY
In an Italian study, 8 of 15 (53%) chronic obstructive pulmonary disease (COPD) patients treated with 3.0 g of ritipenem acoxil developed mild to severe gastrointestinal complaints (diarrhea). Few other toxicity data are currently available.
CLINICAL USES
Ritipenem has been used in a limited number of clinical conditions in which the use of a carbapenem that can be administered both parenterally and orally was considered a possible advantage. Ritipenem is not currently licensed for clinical use by the US FDA. Only a few clinical studies have assessed the role of ritipenem therapy, and these have been in the treatment of respiratory tract or urinary tract infections, but results appear generally disappointing.