Exploring the Versatile Applications and Synthesis of 1,3-Dimethyl-1,3-diphenylurea in Modern Chemistry
Introduction
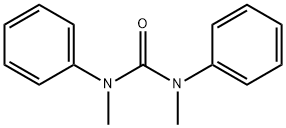
1,3-Dimethyl-1,3-diphenylurea is a specialized chemical compound that has garnered attention in various chemical research and industrial applications. Its unique structure, comprising two phenyl groups attached to a urea backbone modified by methyl groups, allows it to exhibit distinct physical and chemical properties. This molecular configuration enhances its functionality in a range of chemical reactions, contributing to its versatility and utility in organic synthesis. The presence of the phenyl groups increases its hydrophobic nature, which can be crucial for applications requiring non-polar properties. As such, this compound plays a pivotal role in developing advanced materials and pharmaceutical agents[1].

Figure 1 Characteristics of 1,3-Dimethyl-1,3-diphenylurea
Synthesis Methods
The synthesis of 1,3-Dimethyl-1,3-diphenylurea primarily involves the reaction of dimethylamine with diphenylcarbamoyl chloride. This process typically follows a stepwise mechanism where diphenylcarbamoyl chloride is first treated with dimethylamine in a suitable solvent like dichloromethane. The reaction is conducted under controlled temperatures and often requires the presence of a base such as triethylamine to capture the hydrochloric acid formed as a byproduct.
To optimize yield and purity, the reaction mixture may undergo various refinement steps including recrystallization and chromatography. These methods ensure that the final product is free from impurities and suitable for practical applications. Advanced synthesis techniques might also be discussed, emphasizing innovations in catalysis and green chemistry principles aimed at improving the efficiency and environmental impact of the production process.
Main Components
Chemically, 1,3-Dimethyl-1,3-diphenylurea is characterized by its unique arrangement of functional groups. The central urea linkage is pivotal, providing the fundamental framework that contributes to its stability and reactivity. Each phenyl group enhances the compound’s organic solubility and potential interactions with various substrates, making it particularly useful in organic synthesis and polymer chemistry.
Further, the electron-donating effect of the methyl groups at the nitrogen atoms modulates the electronic properties of the urea linkage, affecting its hydrogen bonding capabilities and thermal stability. These attributes are critical when the compound is employed in high-temperature applications or as a stabilizing agent.
Applications
1,3-Dimethyl-1,3-diphenylurea finds its applications in a myriad of chemical processes. It is notably used as a catalyst in certain types of polymerizations, where its ability to stabilize reactive intermediates plays a crucial role. Additionally, its properties make it an ideal candidate for use in the production of certain pharmaceuticals, where it functions as an intermediate in the synthesis of more complex molecules. In this context, it aids in enhancing the efficacy and yield of pharmaceutical products.
In materials science, the compound’s thermal stability and organic solubility allow it to be used in the production of specialty plastics and coatings, tailored for high-performance applications. The environmental stability of 1,3-Dimethyl-1,3-diphenylurea also makes it a preferred choice in applications demanding long-term durability. Its chemical resilience under harsh conditions makes it particularly valuable in the automotive and aerospace industries, where materials are subjected to extreme environmental stresses[2].
Precautions
While handling 1,3-Dimethyl-1,3-diphenylurea, it is essential to adhere to strict safety protocols. Due to its chemical nature, the compound can pose health risks if not managed properly. It is advisable to use personal protective equipment, such as gloves and goggles, and ensure that all interactions occur in a well-ventilated area or under a fume hood. Exposure control measures should also include the use of appropriate respiratory protection if there is a risk of inhalation of dust or vapors.
Storage guidelines recommend keeping the compound in a cool, dry place away from direct sunlight and moisture to preserve its stability and prevent degradation. Additionally, proper waste disposal methods should be followed to minimize environmental impact, adhering to local regulations and guidelines for hazardous materials. It is important to ensure that waste is disposed of in a manner that prevents contamination of the environment and adheres to chemical safety standards.
References
[1]Demont E H, Bamborough P, Chung C, et al. 1, 3-Dimethyl benzimidazolones are potent, selective inhibitors of the BRPF1 bromodomain[J]. ACS medicinal chemistry letters, 2014, 5(11): 1190-1195.
[2]Emery, R., et al. "Conformational preferences in model antiviral compounds: A spectroscopic and computational study of phenylurea and 1, 3-diphenylurea."Physical Chemistry Chemical Physics6.10 (2004): 2816-2820.
Related articles And Qustion
Lastest Price from 1,3-Dimethyl-1,3-diphenylurea manufacturers
US $1.00/g2024-10-23
- CAS:
- 611-92-7
- Min. Order:
- 300g
- Purity:
- 99.8%
- Supply Ability:
- 20 TONS

US $11.90-10.80/KG2024-05-10
- CAS:
- 611-92-7
- Min. Order:
- 1000KG
- Purity:
- 99%
- Supply Ability:
- 200000