1,5-Diaminopentane: Metabolism and Systems Metabolic Engineering
General Description
The metabolism of 1,5-diaminopentane in bacteria, particularly E. coli, involves a biosynthetic pathway branching off the tricarboxylic acid cycle. The synthesis requires pyruvate, ammonium molecules, and NADPH, proceeding through ten steps. Lysine decarboxylase variants, CadA and LdcC, facilitate the final conversion from lysine to 1,5-diaminopentane. Systems metabolic engineering in E. coli involved genetic modifications to enhance production, achieving improved yields. Similarly, C. glutamicum was engineered using lysine decarboxylase from E. coli and metabolic pathway redesign, leading to superior strains with enhanced 1,5-diaminopentane production. These advancements demonstrate the potential for sustainable 1,5-diaminopentane production from renewable sources through systems metabolic engineering.

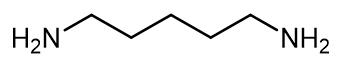
Figure 1. 1,5-Diaminopentane
Metabolism
The metabolism of 1,5-diaminopentane has been extensively studied in bacteria, particularly in E. coli. 1,5-Diaminopentane is synthesized via the lysine pathway, branching off the tricarboxylic acid cycle and starting from oxaloacetate. The biosynthetic pathway requires pyruvate, ammonium molecules, and nicotinamide adenine dinucleotide phosphate (NADPH) molecules, proceeding through ten successive steps. Diaminopimelate, an essential component of cell wall peptidoglycan, is a branch point intermediate directed to the lysine branch. Lysine biosynthesis in E. coli is regulated by various genes and feedback inhibition. The final conversion from lysine to 1,5-Diaminopentane involves lysine decarboxylase variants, CadA and LdcC, both requiring pyridoxal phosphate as a cofactor. The expression of CadA is induced at low pH in the presence of lysine and under anaerobic conditions, leading to 1,5-Diaminopentane excretion, which neutralizes the extracellular medium pH. The expression of CadA is positively regulated by the gene product of the cadC gene and negatively regulated by the product of the cadR gene. As a result, 1,5-Diaminopentane is effectively absent in E. coli growing in minimal medium at neutral pH. 1
Systems metabolic engineering
E. coli
The industrial use of E. coli as a cell factory for 1,5-diaminopentane production has been achieved through systems metabolic engineering. In this approach, various genetic modifications were made to enhance the production of 1,5-diaminopentane. Firstly, a lacI-deleted E. coli W3110 strain was used as a host to enable strong constitutive expression of target genes under the tac promoter. The cadA gene, which encodes lysine decarboxylase, was overexpressed using a multi-copy plasmid under the control of the tac promoter. This allowed for increased conversion of L-lysine to 1,5-diaminopentane. To redirect carbon flux from the threonine pathway to the lysine pathway, enzymes involved in the lysine branch, such as DapA, DapB, and LysA, were overexpressed using the trc promoter integrated into the chromosome. Furthermore, the C. glutamicum ddh gene, responsible for diaminopimelate dehydrogenase activity, was heterologously expressed to achieve direct conversion of tetrahydropicolinate to diaminopimelate. Integration of ddh into the chromosome also increased the expression of glyoxylate enzymes and the supply of oxaloacetate. 1,5-diaminopentane utilization and degradation pathways were disrupted by deleting genes involved in 1,5-diaminopentane degradation and inactivating the putrescine importer PuuP. The final engineered strain, XQ56, exhibited improved 1,5-diaminopentane production, reaching 0.13 g per gram of glucose consumed. Overall, systems metabolic engineering of E. coli successfully enhanced 1,5-diaminopentane production through genetic modifications and pathway manipulations. 2
C. glutamicum
1,5-diaminopentane, a compound derived from lysine by decarboxylation, shows potential for production in C. glutamicum, a strain known for its L-lysine production capabilities. Initially, a wild type strain was modified by incorporating lysine decarboxylase (cadA) from E. coli, resulting in small-scale 1,5-diaminopentane secretion. To enhance production, further modifications included stronger cadA expression and release of lysine pathway feedback inhibition, leading to strains with improved 1,5-diaminopentane yields. Systems metabolic engineering strategies were then employed, such as redesigning key metabolic pathways, optimizing precursor supply, and increasing biosynthetic flux, resulting in superior strains like DAP3a and DAP-3c with enhanced 1,5-diaminopentane production. Additionally, the identification and elimination of unwanted by-products further improved production efficiency. Moreover, advancements in substrate utilization, like utilizing xylose from lignocellulosic biomass, have expanded production possibilities, highlighting the potential for sustainable 1,5-diaminopentane production from renewable sources. These developments showcase the progress in systems metabolic engineering of C. glutamicum for efficient 1,5-diaminopentane production. 2
Reference
1. Kind S, Wittmann C. Bio-based production of the platform chemical 1,5-diaminopentane. Appl Microbiol Biotechnol. 2011;91(5):1287-1296.
2. Chae TU, Ahn JH, Ko YS, et al. Metabolic engineering for the production of dicarboxylic acids and diamines. Metab Eng. 2020;58:2-16.
Related articles And Qustion
Lastest Price from 1,5-Diaminopentane manufacturers

US $10.00/KG2025-04-21
- CAS:
- 462-94-2
- Min. Order:
- 1KG
- Purity:
- 99%
- Supply Ability:
- 100 mt

US $10.00/KG2024-10-11
- CAS:
- 462-94-2
- Min. Order:
- 1KG
- Purity:
- 99.%
- Supply Ability:
- 10 ton