Recombinant Human Vaspin
Recombinant Human Vaspin
¥646
5μg
起订
¥1613
25μg
起订
¥5925
100μg
起订
¥18967
1mg
起订
上海 更新日期:2026-05-13
产品详情:
- 英文名称:
- Recombinant Human Vaspin
- 保存条件:
- -20℃或更低温度保存,至少一年有效。由于蛋白的每次冻融均会引起部分失活,所以首次配制成相应浓度的储存液后(请根据产品简介中Reconstitution一栏的信息配制储存液),须分装后-20℃或更低温度冻存,以避免反复冻融。
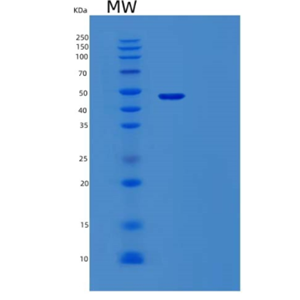
- 纯度规格:
- 99.99%
- 产品类别:
- 分子生物试剂 多肽与蛋白
公司简介
上海康朗生物科技有限公司是一家集研发、生产、销售、服务于一体的一家生物科技企业,专营生化试剂、标准品、基因、蛋白、抗体、Elisa试剂盒、细胞生物学,分子生物学等高生物产品。总部位于上海,并在北京、广东,江西,吉林等全国30多个省市设有分公司和代理机构。涉及的产品被中国科学院、清华、北大、复旦,上海交大,复旦医学院,上海中医药大学,华东师范大学,第二军医大学,曙光医院,浦东新区人民医院等知名科研院所广泛使用,可靠而稳定的质量和完善的售后服务确。
| 成立日期 | (11年) |
| 注册资本 | 100万元整 |
| 员工人数 | 50-100人 |
| 年营业额 | ¥ 100万以内 |
| 经营模式 | 工厂 |
| 主营行业 | 中间体,化学试剂,医药原料 |
Recombinant Human Vaspin相关厂家报价
-
- Recombinant Human Vaspin Protein
- 上海沪震实业有限公司 VIP
- 2026-05-13
- ¥1950
-
- Recombinant Human Vaspin
- 碧云天生物技术有限公司
- 2026-05-07
- 询价
-

- 重组 人Vaspin 蛋白 生产供应商 艾普蒂生物
- 湖北艾普蒂生物工程有限公司
- 2024-02-21
- 询价
-
- Recombinant Human Vaspin
- 碧云天生物技术有限公司
- 2026-05-07
- 询价
-
- Recombinant Human Vaspin
- 碧云天生物技术有限公司
- 2026-05-07
- 询价
-
- Recombinant Human Vaspin
- 碧云天生物技术有限公司
- 2026-05-07
- 询价


