(Bococizumab)Biosimilar Reference Antibody-GS40094
Bococizumab
¥300
100μg
起订
湖北 更新日期:2026-05-07
产品详情:
- 英文名称:
- Bococizumab
- CAS号:
- 1407495-02-6
- 品牌:
- Mabnus
- 产地:
- 武汉
- 保存条件:
- store at -20°C or -80°C. Avoid repeated freeze.
- 纯度规格:
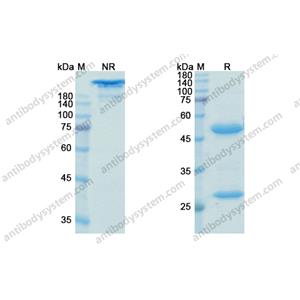
- >98% by SDS-PAGE
- 重组:
- 是
- 应用:
- ELISA;FC;Functional Assays;IF
- 种属反应性:
- Human
- 宿主:
- Humanised
- 偶联物:
- 否
- 靶点:
- PCSK9
- 免疫原:
- PCSK9
- 亚型:
- IgG2 - kappa
- 验证方法:
- (Bococizumab)Biosimilar Reference Antibody(GS40094) on SDS-PAGE under non-reducing (NR) and reducing (R) condition.
公司简介
武汉迈思生物科技有限公司立足于解决行业内功能活性蛋白表达及高亲和力、高灵敏度抗体开发难的痛点,依托自建的两大核心技术平台:哺乳动物细胞蛋白表达平台和单个B细胞抗体开发平台,竭诚为广大科研机构、高等院校、IVD企业、医药企业等提供一站式蛋白表达及抗体开发相关产品和技术服务。
| 成立日期 | (5年) |
| 注册资本 | 壹佰伍拾万圆人民币 |
| 员工人数 | 10-50人 |
| 年营业额 | ¥ 1000万-5000万 |
| 经营模式 | 工厂,试剂,定制,服务 |
| 主营行业 | 抗体,蛋白组学 |
Bococizumab相关厂家报价
-

- Globotriose (Gb3) Analytical Reference 赛可锐 现 货
- 上海赛可锐生物科技有限公司 VIP
- 2026-05-12
- 询价
-
- Reference 2(单道,20-200μl)
- 碧云天生物技术有限公司
- 2026-05-07
- 询价
-
- Eppendorf Reference 单道可调移液器
- 北京索莱宝科技有限公司 VIP
- 2026-05-13
- ¥2150.00
-
- 伯考赛珠单抗,Bococizumab,anti-PCSK9 antibody 抗体
- 武汉佰乐博生物技术有限公司 VIP
- 2026-05-13
- 询价
-

- 伯考赛珠单抗|T76739|TargetMol
- TargetMol中国(陶术生物) VIP
- 2026-04-02
- ¥3180





