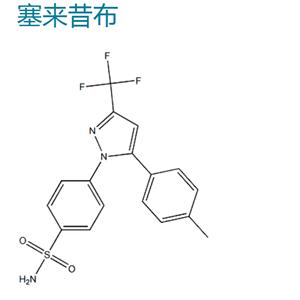
塞来昔布|T0466
Celecoxib
169590-42-5
169590-42-5
¥228
25mg
起订
¥345
50mg
起订
¥541
100mg
起订
上海 更新日期:2026-05-08
产品详情:
- 中文名称:
- 塞来昔布
- 英文名称:
- Celecoxib
- CAS号:
- 169590-42-5
- 品牌:
- TargetMol
- 产地:
- 美国
- 保存条件:
- Powder: -20°C for 3 years | In solvent: -80°C for 1 year Shipping with blue ice/Shipping at ambient temperature.
- 纯度规格:
- 99.89%
- 产品类别:
- 抑制剂
- 货号:
- T0466
公司简介
上海陶术生物科技有限公司为美国Target Molecule Corp. ( Target Mol ) 在上海建立的全资子公司。我们与美国波士顿、德国慕尼黑的同事一起,为北美、欧洲和亚洲从事药物研发和生物学研究的科学家提供优质的产品和专业的服务。公司下设筛选事业部,化学事业部,生物事业部和新材料部。
从虚拟筛选到实体化合物分子供应;从商业化产品销售到个性化定制合成;从对明确靶点的分子筛选到对明确分子的多靶点筛选,从高通量筛选到化学结构优化,我们都可以满足您的科研用品及技术服务的需求。
经过在中国市场五年的精心耕耘,我们已成为筛选化合物领域优秀的供应商,为超过五百家学校和各类企业提供了品质卓越的小分子化合物和药物筛
| 成立日期 | (14年) |
| 注册资本 | 566.2651万人民币 |
| 员工人数 | 100-500人 |
| 年营业额 | ¥ 1亿以上 |
| 经营模式 | 贸易,试剂,定制,服务 |
| 主营行业 | 化学试剂,生物活性小分子 |
塞来昔布相关厂家报价 更多
-
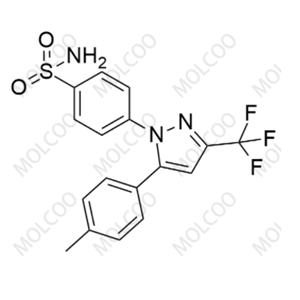
- 塞来昔布
- 深圳摩科生化科技有限公司 VIP
- 2026-05-14
- ¥515
-

- 塞来昔布 169590-42-5
- 湖北威德利化学科技有限公司 VIP
- 2026-05-14
- ¥500
-

- 塞来昔布,169590-42-5,抗炎镇痛、小分子抑制剂,湖北源梦
- 湖北源梦生物科技有限公司 VIP
- 2026-05-14
- ¥280
-
- 塞来西布
- 河南开研生物科技有限公司 VIP
- 2026-05-14
- 询价
-

- 塞来昔布原料;99.8%塞来昔布原粉
- 湖北鸿运隆生物科技有限公司 VIP
- 2026-05-14
- 询价
-

- 塞来昔布169590-42-5
- 天津茂田科技有限公司 VIP
- 2026-05-14
- 询价
-
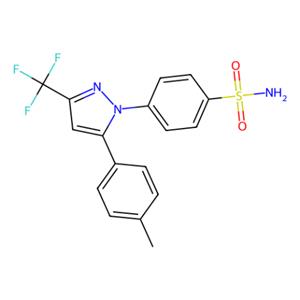
- aladdin 阿拉丁 C129279 塞来昔布 169590-42-5 ≥99%
- 上海阿拉丁生化科技股份有限公司 VIP
- 2026-05-14
- ¥39.90
-
- 噻利考西
- 武汉裕清嘉衡药业有限公司 VIP
- 2026-05-14
- 询价
-

- 塞来昔布 169590-42-5 99%
- 山东迪贝克生物工程有限公司 VIP
- 2026-05-13
- 询价
-

- 塞内昔布—169590-42-5
- 湖北魏氏化学试剂股份有限公司 VIP
- 2026-05-13
- ¥287



