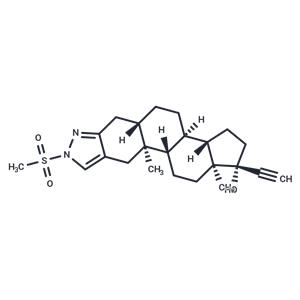
| In vivo | The effects of the steroidal androgen receptor antagonist zanoterone (WIN 49596) and the steroidal 5 alpha-reductase inhibitor finasteride (MK-906) either alone or in combination on prostatic size, histomorphology, and biochemistry were determined in the intact male dog. Additionally, the effects of treatment with zanoterone and/or finasteride on testicular size, serum testosterone and LH levels, and spermatogenesis were determined in the same dogs. Daily oral treatment for 16 weeks with either zanoterone alone at 10 mg/kg.day or finasteride alone at 1.0 mg/kg/day reduced (P < 0.05) the size of the prostate, resulting in mild to moderate diffuse glandular atrophy of the prostate, and decreased prostatic DNA and prostatic arginine esterase (the primary canine prostatic protein) levels compared to those in intact controls. These changes occurred with no effect on testicular weight, testicular histomorphology, daily sperm production, or serum LH levels. Serum testosterone concentrations were increased (P < 0.05) approximately 3-fold in the 10 mg/kg/day zanoterone treatment group compared to those in intact controls. Combination treatment of male dogs for 16 weeks with zanoterone (10 mg/kg/day) plus finasteride (1.0 mg/kg.day) orally also reduced (P < 0.05) prostate size, resulted in moderate to marked diffuse prostatic glandular atrophy, and decreased prostatic DNA and arginine esterase levels more than either drug alone, without affecting testicular size, testicular histomorphology, serum LH concentrations, or serum testosterone concentrations compared to those in intact controls. The effects of combination treatment with zanoterone and finasteride on prostatic size; histomorphology; and DNA, arginine esterase protein, and arginine esterase mRNA levels were similar to those observed in castrate controls. In addition, in situ estimates of prostatic size using transrectal ultrasonography indicated that the median time to 70% prostatic regression in dogs administered a combination of zanoterone plus finasteride was similar to that in castrate controls (9.6 and 9.3 weeks, respectively), indicating that the combination was more effective in causing prostatic regression than either drug alone. Finally, at the dosages used, no adverse effects of combination treatment with zanoterone plus finasteride on testicular or other major body organ weights were observed. Based on these results, combination therapy using zanoterone and finasteride for the treatment of human androgen-dependent disorders such as benign prostatic hyperplasia and prostate cancer has potential utility.[1] |

 United States
United States