| CAS: | 104-98-3 |
| MF: | C6H6N2O2 |
| MW: | 138.12 |
| EINECS: | 203-258-4 |
| Product Categories: | Imidazoles, Pyrroles, Pyrazoles, Pyrrolidines;Heterocyclic Compounds |
| Mol File: | 104-98-3.mol |
 |
|
| Urocanic acid Chemical Properties |
| Melting point | 226-228 °C(lit.) |
| Boiling point | 253.51°C (rough estimate) |
| density | 1.3471 (rough estimate) |
| refractive index | 1.5100 (estimate) |
| storage temp. | Keep in dark place,Sealed in dry,2-8°C |
| solubility | 1.5g/l |
| pka | 2.94±0.10(Predicted) |
| form | Powder |
| color | White to beige |
| Water Solubility | SLIGHTLY SOLUBLE |
| BRN | 81405 |
| InChI | InChI=1S/C6H6N2O2/c9-6(10)2-1-5-3-7-4-8-5/h1-4H,(H,7,8)(H,9,10) |
| InChIKey | LOIYMIARKYCTBW-UHFFFAOYSA-N |
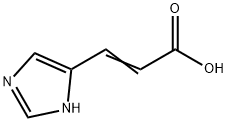
| SMILES | C(O)(=O)C=CC1NC=NC=1 |
| CAS DataBase Reference | 104-98-3(CAS DataBase Reference) |
| EPA Substance Registry System | 2-Propenoic acid, 3-(1H-imidazol-4-yl)- (104-98-3) |
| Urocanic acid Usage And Synthesis |
| Chemical Properties | White to beige fine powder. |
| Uses | Urocanic Acid is a biomarker in the fecal metabolic profiling of breast cancer patients. |
| Definition | ChEBI: Urocanic acid is an alpha,beta-unsaturated monocarboxylic acid that is prop-2-enoic acid substituted by a 1H-imidazol-4-yl group at position 3. It is a metabolite of hidtidine. It has a role as a chromophore and a human metabolite. It is an alpha,beta-unsaturated monocarboxylic acid and a member of imidazoles. It is a conjugate acid of a urocanate. |
| Biological Functions | Urocanic acid, produced in the upper layers of mammalian skin, is a major absorber of ultraviolet radiation (UVR). Urocanic acid (UCA) is formed in the upper layers of the epidermis where filaggrin, a histidine-rich filamentous protein produced after caspase-14 cleavage of profilaggrin, is broken down by proteinases into component amino acids. |
| General Description | 4-Imidazoleacrylic acid also known as urocanic acid is a natural metabolite derived from histidine. It is majorly used as a UV chromophore with a strong absorption spectrum in the UV-B region in the range of 300-280 nm. |
| Synthesis | A method of producing trans-urocanic acid, which involves treating D-glucose with aqueous ammonia and formalin in molar ratio D-glucose: aqueous ammonia: formalin equal to 1:24:2.7, in water in the presence of basic copper carbonate at temperature 85-90°C for 3 hours to form (1S,2S,3S)-1-(1H-imidazol-4-yl)-butane-1,2,3,4-tetrazole, which is extracted in form of a hydrochloride. Through periodate splitting in water, the obtained (1S,2S,3S)-1-(1H-imidazol-4-yl)-butane-1,2,3,4-tetrazole is converted at room temperature to 1H-imidazole-4-carbaldehyde, condensation of which, in acetic anhydride in the presence of anhydrous potassium acetate at temperature 120°C for 2 hours, leads to formation of trans-urocanic acid with subsequent extraction thereof from the reaction mass. |
| Purification Methods | Crystallise the acid from water and dry it at 100o. The trans-isomer [3465-72-3] has m 225o (229-230o, 230-231o or 231o(dec, from H2O) and pK1 3.5 and pK2 5.6, and the picrate has m 225o(dec, from H2O). The cis-isomer [7699-35-6] has m 175-176o (178-179o or 180-184o dec, from H2O) and pK1 3.0 and pK2 6.7, and the picrate has m 204o (from H2O). [Beilstein 25 H 124, 25 I 536, 25 II 121, 25 III/IV 786.] |
Packing &shipping&Payment
Packing:25kg/drum
Shipping:by sea or by air
Payment:T/T,western union,moneygram
Packaging Details drum
Port:Tianjin
Lead Time :
| Quantity(Kilograms) | 1 - 10000 | >10000 |
| Est. Time(days) | 5 | To be negotiated |

 Company information
Company information
Hebei Mojin Biotechnology Co., Ltd, Our company is a professional in 4'-Methylacetophenone,Levamisole hydrochloride ,N-Methylformamide and other chemical reagents research and development production enterprises. Our business covers more than 30 countries, most of the big customers come from Europe, America and other countries in the world, we can guarantee the quality and price. In recent decades, with the efforts of all employees, we have established many cooperative companies in shandong, henan, guangdong and other places. Our corporate purpose is based on the market, enhance the strength, take the road of scientific and environmental sustainable development, relying on the country. Technology r & d center, increase the investment in r & d, based on the domestic market, expand the international market, manufacturing quality products, sincere service to the society, into a modern, ecological, scientific and technological enterprise world.
 Advantage
Advantage
In stock