N-nitroso-N-octylglucamine
Product Code:N031281
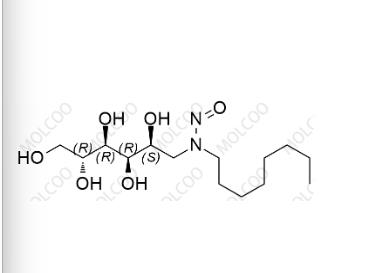
English Name:N-nitroso-N-octylglucamine
English Alias:N-octyl-N-((2S,3R,4R,5R)-2,3,4,5,6-pentahydroxyhexyl)nitrous amide
CAS No.:[Not Available]
Molecular Formula:C₁₄H₃₀N₂O₆
Molecular Weight:322.40
High-Purity Reference Standard:Confirmed by HPLC (≥99.0%), NMR (1H, 13C), HRMS, and circular dichroism for structure, suitable for precise analysis of N-nitroso impurities.
Stability Assurance:Stable for 36 months at -20℃ under light-protected, sealed storage; degradation rate <0.2% in acetonitrile-water solution within 6 months.
Quality Control Testing:Used for UPLC-MS/MS detection of N-nitroso impurities in meglumine-containing drugs (e.g., contrast agents), controlling content to meet ICH M7 standards (≤1.5 μg/day).
Process Optimization Research:Monitors N-nitroso-N-octylglucamine formed by nitrosation side reactions during meglumine derivative synthesis or storage, reducing generation by >60% by adjusting reaction temperature (e.g., 5-10℃) and antioxidant dosage.
Method Validation:Serves as a standard for developing nitrosamine impurity detection methods, verifying UPLC resolution (≥3.0) and LOD (0.005 ng/mL).
N-nitroso compounds (NOCs) are potential carcinogens prevalent in pharmaceuticals, food, and the environment. N-nitroso-N-octylglucamine, as an N-nitroso impurity of meglumine compounds, may form during drug synthesis via reactions between meglumine and nitrites or through spontaneous nitrosation under acidic or high-temperature storage conditions. Its nitrosamide group is genotoxic. With stricter FDA and EMA regulations on genotoxic impurities (GTIs), studying such impurities is crucial for ensuring drug safety.
Detection Technology:UPLC-MS/MS with C18 column (1.7μm) and 0.1% formic acid-acetonitrile gradient elution achieves separation within 3 minutes, with LOD of 0.002 ng/mL for trace nitrosamine analysis.
Formation Mechanism:Formed by reaction of octyl meglumine with sodium nitrite under acidic conditions (pH<4); optimizing reaction pH (neutral) and avoiding nitrite additives inhibits its formation.
Safety Evaluation:In vitro Ames tests show mutagenicity; the permitted daily intake (TTC value) is determined as 1.5 μg/day, requiring strict content control. Accelerated stability testing is ongoing to monitor nitrosation rates under different humidity and light conditions.
This product is intended for laboratory use only!
WhatsAPP: +86 17320513646
E-mail: anna@molcoo.com
NEW IN STOCK!
The Molcoo Laboratory added drug impurity reference standards, including Baricitinib, Piperazine, Benzylpenicillin, Tranilast and multiple N-Nitroso drug impurities! Now available for immediate delivery!