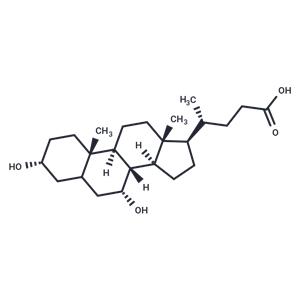
| Name | Chenodeoxycholic acid |
| Description | Chenodeoxycholic acid (CDCA) is a bile acid that inhibits 11β-HSD2 with an IC50 value of 22 mM. Chenodeoxycholic acid acts as a detergent to dissolve fat for intestinal absorption and is reabsorbed by the small intestine. Chenodeoxycholic acid is used as a choledocholithiasis, choledocholithiasis and laxative, as well as to prevent or dissolve gallstones. |
| Cell Research | The cell viability is analyzed by incubating transfected HEK-293 cells and CHO cells for 1 h with the corresponding concentration of bile acid and staining with trypan blue. The toxicity of bile acids is analyzed using the tetrazolium salt MTT (3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide) according to the cell proliferation kit I. No significant differences between control and bile acid-treated cells are obtained in both tests. |
| Kinase Assay | Briefly, transfected HEK-293 cells, incubated in charcoal-treated Dulbecco's modified Eagle's medium for 24 h, are washed once with Hanks' solution and resuspended in a buffer containing 100 mM NaCl, 1 mM MgCl2, 1 mM EDTA, 1 mM EGTA, 250 mMsucrose, 20 mM Tris-HCl, pH 7.4. Cells are lysed by freezing in liquid nitrogen. Dehydrogenase activity is measured in a final volume of 20 μL containing the appropriate concentration of bile acid, 30 nCi of [3H]cortisol, and unlabeled cortisol to a final concentrations of 50 nM. The reaction is started by mixing cell lysate with the reaction mixture. Alternatively, endoplasmic reticulum microsomes are prepared from transfected HEK-293 cells and incubated with reaction mixture containing various concentrations of cortisol and CDCA. Incubation proceeded for 20 min, and the conversion of cortisol to cortisone is determined by thin layer chromatography (TLC). Because of the inaccuracy of the TLC method at low conversion rates and the end-product inhibition of 11βHSD2 at conversion rates higher than 60-70%, only conversion rates between 10 and 60% are considered for calculation. The inhibitory constant IC50 is evaluated using the curve-fitting program. Results are expressed as means±S.E. and consist of at least four independent measurements. |
| In vitro | METHODS: Caco-2 cells were treated with Chenodeoxycholic acid (0.1-1000 μM) for 24 hours, and cell growth inhibition was measured by MTT assay.
RESULTS: Chenodeoxycholic acid significantly inhibited Caco-2 cell growth (IC50=106 μM). [1] |
| Storage | Powder: -20°C for 3 years | In solvent: -80°C for 1 year
Shipping with blue ice/Shipping at ambient temperature. |
| Solubility Information | Ethanol : 79 mg/mL (201.24 mM), Sonication is recommended.
DMSO : 100 mg/mL (254.73 mM), Sonication is recommended.
H2O : < 1 mg/mL (insoluble or slightly soluble)
|
| Keywords | PotassiumChannel | Potassium Channel | NR1H4 | Inhibitor | inhibit | FXR | EndogenousMetabolite | Endogenous Metabolite | Chenodeoxycholic acid | Bile acid receptor | Autophagy |
| Inhibitors Related | Sucrose | Aceglutamide | Hemin | Nicotinamide riboside malate | DL-Lysine | D(+)-Raffinose pentahydrate | Guanidine hydrochloride | Malic acid | Glycerol | Thymidine | Corn starch | Gluconate Calcium |
| Related Compound Libraries | Anti-Tumor Natural Product Library | Pain-Related Compound Library | Bioactive Compound Library | Membrane Protein-targeted Compound Library | EMA Approved Drug Library | Anti-Cancer Clinical Compound Library | Microbial Natural Product Library | Natural Product Library for HTS | FDA-Approved Drug Library | Human Endogenous Metabolite Library | Gut Microbial Metabolite Library | Anti-Cancer Drug Library |

 United States
United States