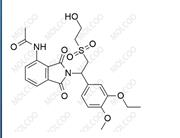
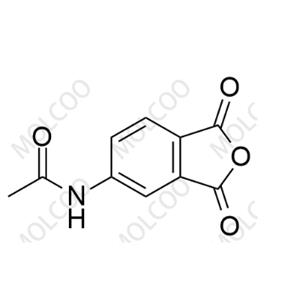
Apremilast Impurity;22235-04-7

WhatsAPP: +86 17386083646
E-mail: anna@molcoo.com
Product Code:A017082
English Name:Apremilast Impurity 82
English Alias:N-(1,3-dioxo-1,3-dihydroisobenzofuran-5-yl)acetamide
CAS No.:22235-04-7
Molecular Formula:C₁₀H₇NO₄
Molecular Weight:205.17
High-Purity Reference Standard:Confirmed by HPLC (≥99.0%), NMR (1H, 13C), HRMS, and elemental analysis, suitable for Apremilast impurity analysis and quality control.
Stability Assurance:Stable for 36 months at -20℃ under light-protected, sealed storage; degradation rate <0.3% in common solvents like methanol and acetonitrile within 6 months.
Quality Control Testing:Used for UPLC-MS/MS detection of Impurity 82 in Apremilast API and formulations, controlling content to meet ICH Q3A standards (single impurity limit ≤0.1%).
Process Optimization Research:Monitors impurity formation during Apremilast synthesis, reducing generation by >30% by adjusting acylation temperature (e.g., 50-60℃) and reaction time.
Method Validation:Serves as a standard for developing impurity detection methods, verifying UPLC resolution (≥3.0) and LOD (0.01 ng/mL).
Apremilast, a phosphodiesterase 4 (PDE4) inhibitor, is used for treating psoriatic arthritis and plaque psoriasis. Impurity 82, as a process-related impurity in Apremilast synthesis, may originate from side reactions between phthalic anhydride and acetamide. Its amide and isobenzofurandione groups may affect drug stability and target binding. With stricter requirements for autoimmune disease drug impurities, studying this impurity is crucial for ensuring drug quality.
Detection Technology:UPLC-MS/MS with C18 column (1.7μm) and 0.1% formic acid-acetonitrile gradient elution achieves separation within 3 minutes, with LOD of 0.005 ng/mL for trace impurity analysis.
Formation Mechanism:Formed by condensation of phthalic anhydride with acetamide under alkaline conditions (e.g., sodium carbonate/methanol system); optimizing reaction pH and raw material ratio inhibits side reactions.
Safety Evaluation:In vitro cytotoxicity shows IC₅₀ of 189.3 μM against HaCaT cells (Apremilast IC₅₀=10.8 μM), with lower toxicity than the main drug but requiring strict content control. Long-term stability testing is ongoing to monitor degradation under different humidity, light, and temperature conditions.
NOTE!
We can also customize related analogues and modified peptides including HPLC, MS, 1H-NMR, MS, HPLC, IR, UV, COA, MSDS.
This product is intended for laboratory use only!
WhatsAPP: +86 17386083646
E-mail: anna@molcoo.com
NEW IN STOCK!
The Molcoo Laboratory added drug impurity reference standards, including Baricitinib, Piperazine, Benzylpenicillin, Tranilast and multiple N-Nitroso drug impurities! Now available for immediate delivery!





 China
China