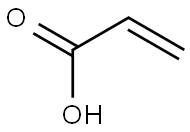
| Product Name | Acrylic acid |
| CAS No | 79-10-7 |
| EC-No | 201-177-9 |
| Min. Order | 1KG |
| Purity | 99% |
| Supply Ability | 50000KG/month |
| Release date | 2025/02/28 |
| CAS: | 79-10-7 |
| MF: | C3H4O2 |
| MW: | 72.06 |
| EINECS: | 201-177-9 |
| Product Categories: | Aliphatics;C1 to C5;Acrylic Acids and Salts;Acrylic Monomers;Carbonyl Compounds;Carboxylic Acids;Industrial/Fine Chemicals;Organic acids;omega-Functional Alkanols, Carboxylic Acids, Amines & Halides;omega-Unsaturated Carboxylic Acids;Monomers;Pyridines ,Heterocyclic Acids;79-10-7 |
| Mol File: | 79-10-7.mol |
 | |
The easiest way to prepare pure acrylic acid in the laboratory is to exchange the ester of formic acid and readily available methyl acrylate. Sulfuric acid is a good catalyst.
CH2=CHCOOCH3 +HCOOH→CH2=CHCOOH+HCOOCH3
Acrolein can be oxidized in the liquid phase at 20-40℃ with silver or vanadium as the catalyst and methoxybenzene as the solvent which glacial acetic acid is more commonly used. It has been reported that the yield of acrylic acid can reach 65-90% based on the consumption of acrolein.
CH2=CHCHO+ 1/2 O2→CH2=CHCOOH
A mixture of water and 2,3 dibromopropionic acid is treated with zinc powder can obtain an aqueous solution of acrylic acid with a yield of 90%.
CH2BrCHBrCOOH +Zn →CH2=CHCOOH+ZnBr2
Acrylic acid also can be obtained by pyrolyzing sec-butyl acrylate at 500℃, or pyrolyzing ethyl acrylate at 570℃.
CH2=CHCOOCH(CH3)CH2CH3 → CH2=CHCOOH+CH3CH=CHCH3
At 200-300℃, acrylic acid can be obtained by passing carbon dioxide and ethylene through the silica gel impregnated with iron sulfite.
CH2=CH2+CO2→CH2=CHCOOH
Acrylic acid and esters are flammable, reactive, volatile liquids based on an alpha-, beta-unsaturated carboxyl structure. Incorporation of varying percentages of acrylate monomers permits the production of many formulations for latex and solution copolymers, copolymer plastics and cross-linkable polymer systems. Their performance characteristics—which impart varying degrees of tackiness, durability, hardness, and glass transition temperatures—promote consumption in many end-use applications. Major markets for the esters include surface coatings, textiles, adhesives, and plastics[8][9].
Polyacrylic acid which produced by acrylic acid can be further modified to produce superabsorbent polymers (SAPs) and other polyacrylic acid homopolymers or copolymers used as detergents, dispersants/antiscalants, anionic polyelectrolytes for water treatment, and rheology modifiers[7][8].
SAPs are cross-linked polyacrylates with the ability to absorb and retain more than 100 times their own weight in liquid. They have experienced very strong growth, primarily in baby diapers (nappies) and incontinence products[7][8].
A new application for SAPs is soaker pads used in food packaging. In 2007, the US Food and Drug Adminstration authorised SAPs in packaging with indirect food contact for poultry, meat, fish, fruit and vegetables[7].
Crude acrylic acid (CAA) is made by the oxidation of propylene. About 55% of the CAA is converted to acrylate esters. The remaining 45% is purified to 98–99.5% purity to glacial acrylic acid (GAA), which, in turn, is converted to polyacrylic acid, which is used to produce superabsorbent polymers (SAPs) and other polyacrylic acid copolymers. In 2016, global glacial acrylic acid consumption was estimated to account for about 45% of total crude acrylic acid consumption, of which 79% was consumed for superabsorbent polymers. Growth in GAA consumption is forecast at about 5% per year during 2016?21. Growth in demand for crude acrylic acid is forecast at 4.5% per year during 2016?21, driven by growth in superabsorbent polymers at 5.5% and acrylate esters at about 4%. SAP growth will be strongest in China and other areas of Asia, but will be much more moderate in the mature regions of North America, Western Europe, and Japan[8].
Product picture

Packing &shipping&Payment
Shipping:by sea or by air
Payment:T/T,western union,moneygram
Packaging Details drum
Port:Tianjin
Lead Time :
| Quantity(Kilograms) | 1 - 10000 | >10000 |
| Est. Time(days) | 5 | To be negotiated |

 Company information
Company information
Hebei Mojin Biotechnology Co., Ltd, Our company is a professional in 4'-Methylacetophenone,Levamisole hydrochloride ,N-Methylformamide and other chemical reagents research and development production enterprises. Our business covers more than 30 countries, most of the big customers come from Europe, America and other countries in the world, we can guarantee the quality and price. In recent decades, with the efforts of all employees, we have established many cooperative companies in shandong, henan, guangdong and other places. Our corporate purpose is based on the market, enhance the strength, take the road of scientific and environmental sustainable development, relying on the country. Technology r & d center, increase the investment in r & d, based on the domestic market, expand the international market, manufacturing quality products, sincere service to the society, into a modern, ecological, scientific and technological enterprise world.
 Advantage
Advantage
In stock
Company Profile Introduction
-








