Chemical Properties
colourless liquid
Chemical Properties
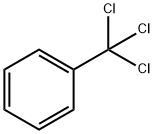
Benzotrichloride is a combustible, colorless to yellow-brown, oily liquid that fumes on contact with air. It has a penetrating odor.
Uses
α,α,α-Trichlorotoluene was used in determination of chlorinated toluenes in effluents of wastewater treatment plants and textile industries by closed loop stripping analysis.
Uses
In dye chemistry. In organic syntheses (source of benzenyl group).
General Description
A clear colorless to yellowish colored liquid with a penetrating odor. Denser than water and vapors are heavier than air. May be toxic by inhalation or ingestion. Burns skin, eyes, and mucous membranes. Insoluble in water. Used to make dyes and other chemicals.
Air & Water Reactions
Hydrolyzes in the presence of moisture. The reaction is rather slow due to low solubility in water.
Reactivity Profile
Benzotrichloride hydrolyzes in the presence of moisture. Benzotrichloride reacts with water, lime, ammonia, strong alkalis, organic amines, chlorates and acids.
Hazard
Highly toxic by inhalation, fumes highly irritant. Eye, skin, and upper respiratory tract irri- tant. Probable carcinogen.
Health Hazard
Toxic by inhalation; fumes are highly irritating to skin and mucous membranes. May cause death or permanent injury after very short exposure to small quantities. A suspected carcinogen.
Fire Hazard
Benzotrichloride may react violently with water. Fire may produce irritating or poisonous gases. Flammable/poisonous gases may accumulate in tanks and hopper cars. Materials may ignite combustibles (wood, paper, oil, etc.). Produces toxic and corrosive fumes when Benzotrichloride reacts with water or steam. Similar to hydrochloric acid; material will react with water or steam to produce toxic and corrosive fumes. Hydrolyzes in presence of water forming benzoic and hydrochloric acids.
Potential Exposure
Benzotrichloride is used as a chemical and dye intermediate; extensively used in the dye industry for the production of Malachite green, Rosamine, Quinoline red, and Alizarin yellow A. It can also be used to produce ethyl benzoate. Commercial grades may contain hydrochloric acid, benzylidene chloride, or benzyl chloride.
Carcinogenicity
Benzotrichloride is reasonably anticipated to be a human carcinogen based on sufficient evidence of carcinogenicity from studies in experimental animals.
Shipping
UN2226 Benzotrichloride, Hazard class: 8; Labels: 8—Corrosive material.
Incompatibilities
Benzotrichloride decomposes on heating, on contact with acids and/or water, producing toxic and corrosive hydrogen chloride and benzoic acid. Reacts violently with strong oxidizers, iron and other metals, alkali and earth alkali metals; bases and organic substances, and may cause fire and explosions. On contact with air it emits toxic and corrosive hydrogen chloride. Attacks many metals in presence of water. Attacks many plastics.
Waste Disposal
Incineration with flammable solvent added in incinerator with afterburner and alkaline scrubber.

 China
China