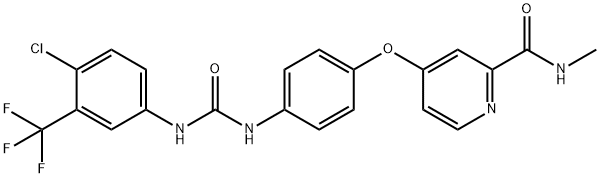
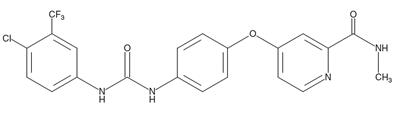
Side effects
The most common adverse reactions (20%), considered to be related to sorafenib, in patients with HCC or RCC are fatigue, weight loss, rash/desquamation, hand-foot skin reaction, alopecia, diarrhea, anorexia, nausea and abdominal pain [12].
Across all tumor types, common side effects (> 10%) include hypertension (9 -13%, grade 4: < 1%; onset: ~ 3 weeks), fatigue (37 -46%), sensory neuropathy (13%), pain (11%), rash/desquamation (19 -40%; grade 3: 1%), handfoot syndrome (21 -30%; grade 3: 6 -8%), alopecia (14 -27%), pruritis (14 -19%), dry skin (10 -11%), hypoalbuminemia (59%), hypophosphatemia (35 -45%; grade 3: 11 -13%; grade 4: < 1%), diarrhea (43 -55%; grade 3: 2 -10%; grade 4: < 1%), lipase increased (40 -41%, usually transient), amylase increased (30 -34, usually transient), abdominal pain (11 -31%), weight loss (10 -30%), anorexia (16 -29%), nausea (23 -24%), vomiting (15 -16%), constipation (14 -15%), muscle pain, weakness, dyspnea (14%), cough (13%) and hemorrhage (15 -18%; grade 3: 2 -3%; grade 4: 2%). Laboratory abnormalities attributable to sorafenib use are also seen and include lymphopenia (23 -47%; grades 3/4: 13%), thrombocytopenia (12 -46%; grades 3/4: 1 -4%), international normalized ration (INR) increased (42%), neutropenia (18%; grades 3/4: 5%), leucopenia, liver dysfunction (11%; grade 3: 2%; grade 4: 1%).
Less frequent side effects (> 1 -10) include cardiac ischemia/infarction (3%), flushing, headache (10%), depression, fever, acne, exfoliative dermatitis, decreased appetite, dyspepsia, dysphagia, esophageal varices bleeding (2%), glossodynia, mucositis, stomatitis, xerostomia, erectile dysfunction, anemia, transaminases increased (transient), joint pain (10%), arthralgia, myalgia, hoarseness and flu-like syndrome.
Rare (< 1%) side effects of sorafenib include acute renal failure, alkaline phosphatase increased, arrhythmia, bilirubin increased, bone pain, cardiac failure, cerebral hemorrhage, congestive heart failure, dehydration, eczema, epistaxis, erythema multiforme, folliculitis, gastritis, gastrointestinal hemorrhage, gastrointestinal perforation, gastrointestinal reflux, gynecomastia, hypersensitivity (skin reaction, urticaria), hypertensive crisis, hyponatremia, hypothyroidism, infection, jaundice, myocardial infarction (MI), mouth pain, myocardial ischemia, pancreatitis, pleural effusion, preeclampsialike syndrome (reversible hypertension and proteinuria), renal failure, respiratory hemorrhage, reversible posterior leukoencephalopathy syndrome (RPLS), rhinorrhea, skin cancer (squamous cell/keratoacanthomas), thromboembolism, tinnitus, transient ischemic attack, tumor lysis syndrome, tumor pain and voice alteration.
Indications
Sorafenib (Nexavar(R), Bayer) was the first approved inhibitor targeting the vascular endothelial growth factor (VEGF) family kinases, which include VEGFR1, VEGR2, and VEGFR3. Sorafenib was originally approved for the treatment of renal cell carcinoma (RCC) in 2005, hepatocellular carcinoma in 2007, and locally recurrent or metastatic thyroid carcinoma refractory to radioactive iodine treatment in 2013. Six other approved inhibitors with VEGFRs as the main targets are sunitinib (Sutent(R), Pfizer) for RCC, soft tissue sarcoma, thyroid cancer,metastatic pancreatic tumors, gastrointestinal stromal tumor, and several other types of carcinomas; pazopanib (Votrient(R), GlaxoSmithKline) for RCC, soft tissue sarcoma, and thyroid cancer; axitinib (Inlyta(R), Pfizer) for RCC,thyroid cancer, and aplastic anemia, as well as T315I-mutant Bcr–Abl1-driven leukemia; regorafenib (Stivarga(R), Bayer) for gastrointestinal stromal tumors and colorectal cancer; nintedanib (Ofev(R), Boehringer Ingelheim) for the non-oncological indication of idiopathic pulmonary fibrosis; and lenvatinib (Lenvima(R), Eisai Inc.) for RCC and different types of thyroid cancers. Sunitinib, pazopanib, and lenvatinib bind to the “DFG-in”conformation of VEGFRs, while axitinib, regorafenib, and nintedanib bind to inactive VEGFRs adopting the “DFG-out”conformation.

 China
China