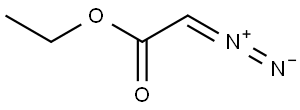
Ethyl diazoacetate
$ 5.00
/5KG
- Min. Order1KG
- Purity99%
- Cas No623-73-4
- Supply Ability1ton
- Update time2019-08-06

career henan chemical co
VIP8Y
 China
China
Enterprise Verified
Business Bank account
Basic Contact Infomation
Business Address
Trade Company



Chemical Properties
| Product Name | Ethyl diazoacetate |
| CAS No | 623-73-4 |
| EC-No | 200-144-5 |
| Min. Order | 1KG |
| Purity | 99% |
| Supply Ability | 1ton |
| Release date | 2019/08/06 |