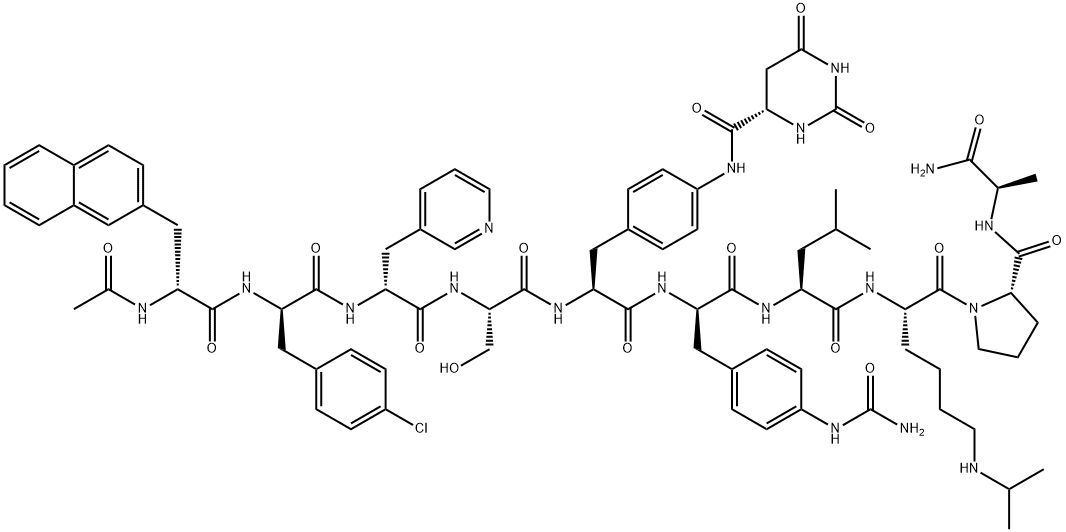
| Description |
Antagonists of GnRH have proven to be an effective therapy for hormonally regulated cancers, such as prostate and some types of breast. As analogs of GnRH, they bind competitively and reversibly to GnRH receptors in the pituitary gland, thereby blocking the release of luteinizing hormone (LH) and follicle-stimulating hormone (FSH). In men, the reduction of LH triggers the ablation of testosterone secretion from the testes, and these castration-like levels have been essential in the effective management of advanced prostate cancer. In comparison to GnRH agonists, antagonists do not suffer from a potential flare of the disease as a result of an initial stimulation of the hypothalamic-pituitary-gonadal axis prior to down-regulation of the GnRH receptor. Moreover, GnRH antagonists provide beneficial effects more rapidly postdosing and result in a more efficient suppression of gonadotropin levels. With this in mind, degarelix acetate has been launched as a third-generation GnRH antagonist for the treatment of prostate cancer, and it joins other third-generation agents, ganirelix and cetronelix, on the market. |
| Originator |
Ferring Pharmaceutical (Switzerland) |
| Uses |
Advanced hormone-dependent prostate carcinoma |
| Brand name |
Firmagon |
| Side effects |
The most common adverse events included injection site reactions (pain, erythema, swelling, or induration), hot flashes, increased weight, and increases in serum levels of transaminases and gamma-glutamyltransferase. In addition to being contraindicated in patients with a previous hypersensitivity to degarelix, it should not be administered to women who are or may become pregnant as fetal harm can occur. Since long-term androgen deprivation therapy prolongs the QT interval, physicians should consider whether the benefits of degarelix outweigh the potential risks in patients with congenital long QT syndrome, electrolyte abnormalities, or congestive heart failure or in patients taking antiarrhythmic medications. |

 China
China