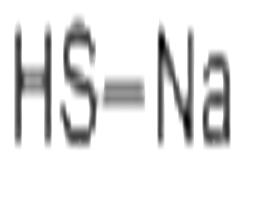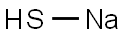| Chemical Properties |
white crystals or powder with a very unpleasant smell |
| Uses |
the copper ore beneficiation,the sulfurous acid dyeing of manmade fibre,the synthesis of organic intermediates,the preparation of auxiliary agent of sulfur dyes,and waste water treatment; raw material to produce ammonium sulphate and ethanethiol |
| General Description |
Sodium hydrosulfide is a colorless to light yellow crystalline solid or fused mass. Sodium hydrosulfide is corrosive to skin and metal. Used in paper pulping, manufacturing dyes, and dehairing hides. |
| Air & Water Reactions |
Soluble in water. The solution slowly evolves hydrogen sulfide gas. Crystals hydrolyze in moist air to sodium hydroxide and sodium sulfide. Liable to spontaneous heating in moist air. |
| Reactivity Profile |
A chemical base. Reacts with acids to release flammable and toxic gaseous hydrogen sulfide. As long as the solution is kept strongly alkaline, pH > 10, there is very little release of H2S. At pH = 7, the percent concentration of H2S released is close to 80%. |
| Health Hazard |
Fire will produce irritating, corrosive and/or toxic gases. Inhalation of decomposition products may cause severe injury or death. Contact with substance may cause severe burns to skin and eyes. Runoff from fire control may cause pollution. |
| Fire Hazard |
Flammable/combustible material. May ignite on contact with moist air or moisture. May burn rapidly with flare-burning effect. Some react vigorously or explosively on contact with water. Some may decompose explosively when heated or involved in a fire. May re-ignite after fire is extinguished. Runoff may create fire or explosion hazard. Containers may explode when heated. |

 China
China