| Description |
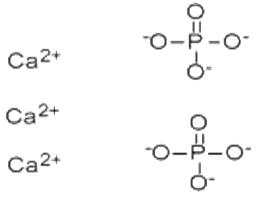
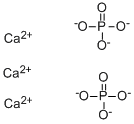
Calcium Phosphate is the calcium salt of phosphoric acid with widely used applications. It occur abundantly in nature in several forms and are the principal minerals for the production of phosphate fertilizers and for a range of phosphorus compounds. For example, the tribasic variety (precipitated calcium phosphate), Ca3(PO4)2, is the principal inorganic constituent of bone ash. The acid salt Ca(H2PO4)2, produced by treating mineral phosphates with sulfuric acid, is employed as a plant food and stabilizer for plastics. It is a natural constituent of mammals, and it is a component of bone replacement transplants in much higher amounts with no toxicological problems.Calcium phosphates are the largest group of artificial bone graft substitutes. This is mainly due to their close resemblance to the mineral components of bone. This Product can be used as a countermeasure for exposure to strontium and radium radionuclides. Upon oral uptake, calcium phosphate competes for and blocks the absorption of radium (Ra-226) and strontium (Sr-90) in the gastrointestinal (GI) tract. |
| Chemical Properties |
white powder |
| Uses |
Tribasic calcium phosphate occurs in nature as minerals, oxydapatite, whitlockite, voelicherite, apatite, phosphorite. It has many industrial applications. Some are similar to the monobasic and dibasic salts. It is used in fertilizers, dental products, ceramics and polishing powder. Some other important applications are in plastics as a stabilizer; as an anticaking agent; as a nutrient supplement in cattle food; for clarifying sugar syrup; as a mordant in dyeing textiles; and as a buffer to control pH. |
| Preparation |
Tribasic calcium phosphate is obtained from naturally occurring minerals for fertilizer applications. The compound may be prepared in the laboratory by the reaction of sodium phosphate with calcium chloride with excess of ammonia. Also, it can be prepared by treatment of calcium hydroxide with phosphoric acid:
2H3PO4 + 3Ca(OH)2 → Ca3(PO4)2 + 6H2O |
| Uses |
manufacture of fertilizers, H3PO4 and P Compounds; manufacture of milk-glass, polishing and dental powders, porcelains, pottery; enameling; clarifying sugar syrups; in animal feeds; as noncaking agent; in the textile industry. |

 China
China