| Description | Regorafenib (BAY 73-4506) is a multi-targeted receptor tyrosine kinase inhibitor with IC50s of 13/4.2/46, 22, 7, 1.5 and 2.5 nM for VEGFR1/2/3, PDGFRβ, Kit, RET and Raf-1, respectively. |
|---|
| Related Catalog | Signaling Pathways >> Autophagy >> Autophagy Signaling Pathways >> Protein Tyrosine Kinase/RTK >> PDGFR Signaling Pathways >> MAPK/ERK Pathway >> Raf Signaling Pathways >> Protein Tyrosine Kinase/RTK >> VEGFR Research Areas >> Cancer |
|---|
| Target | VEGFR1:13 nM (IC50) VEGFR2:4.2 nM (IC50) VEGFR3:46 nM (IC50) PDGFRβ:22 nM (IC50) Raf-1:2.5 nM (IC50) Braf:28 nM (IC50) BRafV600E:19 nM (IC50) |
|---|
| In Vitro | Regorafenib potently inhibits VEGFR2 autophosphorylation in NIH-3T3/VEGFR2 cells with an IC50 of 3 nM. In HAoSMCs, regorafenib inhibits PDGFR-β autophosphorylation after stimulation with PDGF-BB, with an IC50 of 90 nM. Regorafenib inhibits the proliferation of VEGF165-stimulated HUVECs, with an IC50 of 3 nM[1]. Regorafenib causes a concentration-dependent decrease in Hep3B cell growth, having an IC50 of 5 μM. Regorafenib subsequently increases the levels of phospho-c-Jun, a JNK target, but not total c-Jun in Hep3B cells[3]. |
|---|
| In Vivo | Regorafenib effectively inhibits growth of the Colo-205 xenografts in the dose range of 10-100 mg/kg reaching a TGI of 75% at day 14 at the 10 mg/kg dose. In the MDA-MB-231 model, regorafenib is highly efficacious at a dose as low as 3 mg/kg, resulting in a significant TGI of 81%, which increases to 93% at doses of 10 and 30 mg/kg, where tumor stasis is reached[1]. |
|---|
| Kinase Assay | Initial in vitro kinase inhibition profiling is performed at Millipore Corporation at a fixed 1 μM compound concentration under Millipore standard conditions [10 μM adenosine-5′-triphosphate (ATP) concentration]. Inhibitory concentration of 50% (IC50) values are determined from selected responding kinases,e.g., VEGFR1 and RET. TIE2 kinase inhibition is measured with a homogeneous time-resolved fluorescence (HTRF) assay using a recombinant fusion protein of glutathione-S-transferase, the intracellular domain of TIE2 and the peptide biotin-Ahx-EPKDDAYPLYSDFG as substrate. |
|---|
| Cell Assay | For proliferation assays, GIST 882 and TT cells are grown in RPMI medium containing L-glutamine, and MDA-MB-231, HepG2 and A375 cells in DMEM always containing 10% hiFBS. Cells are trypsinized, plated at 5×104 cells/well in 96-well plates in complete media containing 10% FBS and grown overnight at 37°C. The next day, vehicle or regorafenib serially diluted in complete growth media to between 10 μM and 5 nM final concentrations, and 0.2% DMSO, is added and incubation is continued for 96 hr. Cell proliferation is quantified using CellTitre-GloTM. |
|---|
| Animal Admin | Female athymic NCr nu/nu mice, kept in accordance with Federal guidelines, are subcutaneously inoculated with 5×106 Colo-205 or MDA-MB-231 cells or implanted with 1 mm3 786-O tumor fragments. When tumors reach a volume of 100 mm3, regorafenib or vehicle control is administered orally qd×21 in the 786-O model, and qd×9 in the Colo-205 and MDA-MB-231 models, respectively, at doses of 100, 30, 10, and 3 mg/kg. Paclitaxel is administered intravenously at 10 mg/kg in ethanol/Cremophor EL®/saline (12.5%/12.5%/75%) every 2 days×5. Tumor size (volume) is estimated twice weekly (l×w2)/2, and the percentage of tumor growth inhibition (TGI) is obtained from terminal tumor weights (1-T/C×100). Mice are weighed every other day starting from the first day of treatment. The general health status of the mice is monitored daily. |
|---|
| References | [1]. Wilhelm SM, et al. Regorafenib (BAY 73-4506): a new oral multikinase inhibitor of angiogenic, stromal and oncogenic receptor tyrosine kinases with potent preclinical antitumor activity. Int J Cancer, 2011, 129(1), 245-255. [2]. Heng DY, et al. Targeted therapy for metastatic renal cell carcinoma: current treatment and future directions. Ther Adv Med Oncol, 2010, 2(1), 39-49. [3]. Carr BI, et al. Fluoro-Sorafenib (Regorafenib) effects on hepatoma cells: growth inhibition, quiescence, and recovery. J Cell Physiol, 2013, 228(2), 292-297. [4]. Wagner J, et al. Anti-tumor effects of ONC201 in combination with VEGF-inhibitors significantly impacts colorectal cancer growth and survival in vivo through complementary non-overlapping mechanisms. J Exp Clin Cancer Res. 2018 Jan 22;37(1):11. [5]. Matsuoka K, et al. Effective Sequential Combined Chemotherapy with Trifluridine/Tipiracil and Regorafenib in Human Colorectal Cancer Cells. Int J Mol Sci. 2018 Sep 25;19(10). pii: E2915. |
|---|
 China
China
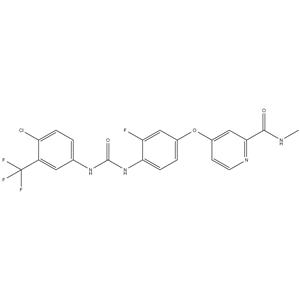

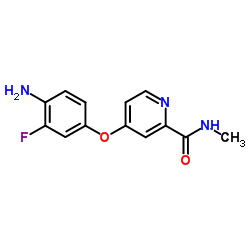
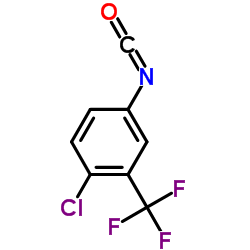
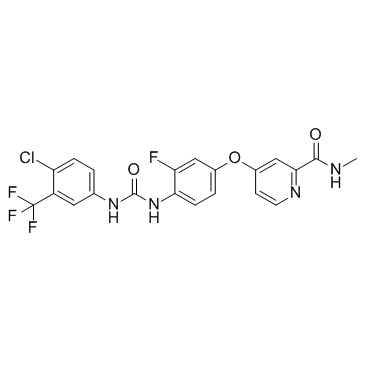
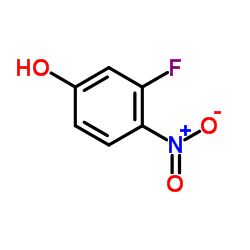
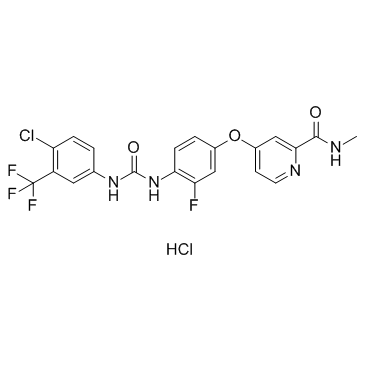 CAS#:835621-07-3
CAS#:835621-07-3