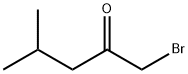
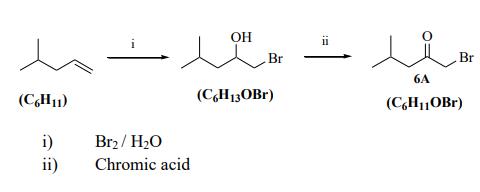
Sytheisis of 1-Bromo-4-methylpentan-2-one: 4-Methylpentene (10.1 g, 120.0 mmmol) was mixed with water (500 mL) at 0 °C. Br2 (19.2 g, 120.0 mmol) was added dropwise and the mixture was stirred overnight at r.t. The mixture was extracted with pet. ether (4 x 75 mL). The organic phase was dried with MgSO4 and the solvent was evaporated under reduced pressure giving the crude product (19.15 g, brown oil). The crude product contained a mixture of 1,2 dibromo-4-methylpentane and 1-bromo-4- methylpentan-2-ol and 2-bromo-4-methylpentanol. The crude product was separated on DFC using the eluent (10% EtOAc in Hexanes) giving 1,2 dibromo-4-methylpentane (11.3 g) and a mixture of 1-bromo-4-methylpentan-2-ol and 2-bromo-4-methylpentanol (1.9 g). Chromic acid solution (12.3 mL) was added to the mixture of 1bromo-4-methylpentan-2-ol and 2- bromo-4-methylpentanol (1. 9 g) was dissolved in diethyl ether (50 mL) and the mixture was stirred overnight at r.t. Saturated NaHCO3 (aq) (200 mL) was added and the mixture was extracted with pet. ether (4 x 75 mL). The organic phase was dried with MgSO4 and the solvent was evaporated under reduced pressure giving the 1-bromo-4-methylpentan-2-one (0.72 g, brown oil, 3.3 %). The yield of this reaction was poor, but the same reaction is reported in 28% yield.
Chromic acid solution was prepared (500 mL):
Na2Cr2O7 x H2O (100 g, 0.33 mol) was dissolved in 300 mL of water. 97 % H2SO4 (136 g, 1. 34 mol) was then added. The solution was then diluted to 500 mL total volume. This solution will oxidize 1.00 mol of secondary alcohol.
Rf = 0.44 (15% EtOAc in Hexanes)