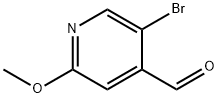
To a methanolic (100 mL) solution of 5-bromo-2-methoxypyridine-4-carbaldehyde (23.5 g, 108.8 mmol) was sequentially added a methanolic (75 mL) solution of iodine (35.9 mg, 141.4 mmol). A methanol (75 mL) solution of potassium hydroxide (15.9 g, 282.8 mmol) was added at 0 °C. The reaction mixture was stirred at 0 °C for 1 h. The reaction was subsequently quenched with saturated aqueous sodium bisulfite solution. The reaction mixture was diluted with dichloromethane (400 mL). The organic phase was separated, washed sequentially with water (150 mL) and brine (150 mL), dried over anhydrous sodium sulfate and concentrated in vacuum. The residue was purified by fast column chromatography (eluent: petroleum ether/ethyl acetate = 20:1, v/v) to afford methyl 5-bromo-2-methoxypyridine-4-carboxylate (14.9 g) as a light yellow solid.