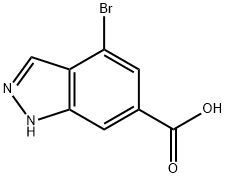
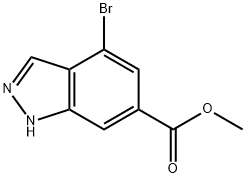
4.2.2 Synthesis of 4-bromo-1H-indazole-6-carboxylic acid (14): 4-bromo-indazole-6-carboxylic acid methyl ester (13) (3.38 g, 13.3 mmol) was dissolved in ethanol (30 mL) and 15% aqueous sodium hydroxide solution (15 mL) was added. The reaction mixture was stirred at room temperature for 4 hours. After completion of the reaction, the solvent was removed by concentration under reduced pressure. The residue was diluted with water and acidified to pH 4 with 1 M hydrochloric acid to precipitate an orange precipitate. The precipitate was collected by filtration and dried under vacuum to give 4-bromo-1H-indazole-6-carboxylic acid (14) (2.64 g, 83%) as a light orange solid. Thin layer chromatography (TLC) Rf value (10% MeOH/CH2Cl2): 0.10. Melting point: 294-296 °C (decomposition).1H NMR (DMSO-d6, δ): 13.78 (1H, broad single peak), 13.3 (1H, broad single peak), 8.15 (2H, single peak), 7.80 (1H, double peak, J = 0.5 Hz).13C NMR (DMSO-d6, δ): 166.4, 139.9, 133.4, 129.9, 125.7, 123.0, 113.0, 111.8. mass spectra (ESI): m/z 240.9 ([M(79Br)+H]+), 242.9 ([M(81Br)+H]+). High Resolution Mass Spectrometry (HRMS, ESI): [M(79Br)+H]+ calculated value 240.9613 (C8H6BrN2O2), measured value 240.9607.