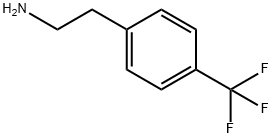
2-(4-Trifluoromethylphenyl)ethylamine is colorless transparent to light yellow liquid at room temperature and pressure, slightly soluble in water, easily soluble in toluene and other organic solvents.
The general procedure for the synthesis of 2-(4-trifluoromethylphenyl)ethylamine from p-trifluoromethylphenylacetonitrile was as follows: (4-trifluoromethylphenyl)acetonitrile (2 g, 10.81 mmol) was dissolved in methanol-ammonia solution (50 mL), which was subsequently added to a Parr hydrogenation reactor that had been charged with nickel Nguyenay (400 mg). The reaction mixture was stirred overnight under hydrogen pressure of 50 psi. The progress of the reaction was monitored by thin layer chromatography (TLC, the unfolding agent was a chloroform solution containing 10% methanol). Upon completion of the reaction, the mixture was filtered through a bed of diatomaceous earth and the filtrate was concentrated and dried under reduced pressure to afford the target product 2-(4-trifluoromethylphenyl)ethylamine (2 g, 97.89% yield). The product was analyzed by LCMS and the purity was 98.73%, m/z = 190.0 ([M+H]+).
[1] Patent: WO2012/35078, 2012, A1. Location in patent: Page/Page column 67
[2] Patent: US2014/45872, 2014, A1. Location in patent: Paragraph 0403
[3] Patent: WO2004/82682, 2004, A1. Location in patent: Page 44-45
[4] Patent: WO2003/93231, 2003, A2. Location in patent: Page/Page column 48
[5] Patent: WO2005/80371, 2005, A1. Location in patent: Page/Page column 49