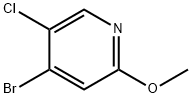
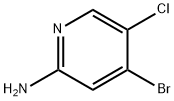
Trimethylchlorosilane (TMS-Cl, 49.0 mL, 386 mmol) was slowly added to methanol (390 mL) at 0 °C. The reaction mixture was gradually warmed to room temperature and stirred continuously for 30 minutes. Subsequently, 4-bromo-5-chloro-2-aminopyridine (27A, 20.0 g, 96.0 mmol) was added to the solution. After the reaction mixture continued stirring for 15 h, sodium nitrite (NaNO2, 2.74 g, 39.8 mmol) was added at room temperature. The reaction mixture was heated to 50 °C and stirred for 3 hours. After completion of the reaction, the mixture was concentrated. The concentrated residue was diluted with ethyl acetate (EtOAc) and the pH was adjusted to 11-13 with 1N aqueous sodium hydroxide solution. extraction was carried out with sodium hydroxide solution (3 times). The organic layers were combined and concentrated. Finally, 4-bromo-5-chloro-2-methoxypyridine (27B) was purified by recrystallization from a solvent mixture of methanol and water to give 4-bromo-5-chloro-2-methoxypyridine (27B) as white acicular crystals (18.0 g, 81.0 mmol, 84% yield).LC-MS analysis: calculated value of 220.92 and measured value of 223.9 for C6H5BrClNO.