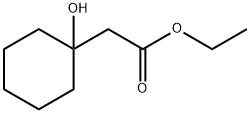
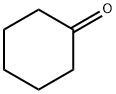
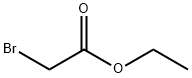
General procedure for the synthesis of ethyl 1-hydroxycyclohexaneacetate from cyclohexanone and ethyl bromoacetate: cf. Example 61; Synthesis of ethyl (1-hydroxycyclohexyl)acetate; Zinc powder (7.6 g, 116.4 mmol) and a small amount of iodine were sequentially added to a solution of THF (100 mL) of cyclohexanone (9.52 g, 97.0 mmol) protected by nitrogen followed by the dropwise addition of bromo ethyl acetate (11.8 mL, 106.7 mmol) was added dropwise. The reaction mixture was heated to reflux for 5 hours. Upon completion of the reaction, 10% sulfuric acid solution (100 mL) was added slowly under ice bath cooling and the mixture was extracted with ethyl acetate. The organic phases were combined, washed with saturated aqueous sodium bicarbonate, dried over anhydrous magnesium sulfate, and concentrated under reduced pressure to give ethyl 1-hydroxycyclohexaneacetate (17.2 g, 94% yield) as a colorless oil.1H-NMR (CDCl3) δ: 1.27 (3H, t, J = 7.0 Hz), 1.35-1.71 (10H, m), 2.46 (2H, s), and 3.43 (1H, s), 4.17 (2H, q, J = 7.4 Hz).