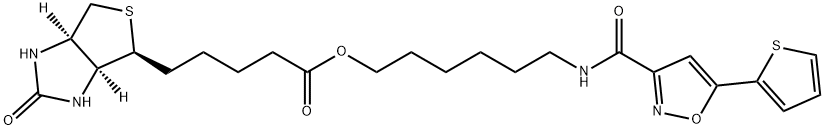
Biotin-isoxazole (1377605-22-5) precipitates hundreds of RNA-binding proteins with significant overlap to the constituents of RNA granules.1,2 In one study roughly 580 nuclear proteins were identified as being precipitated by B-Isox including TAF15, RNA polymerase II (largest subunit), numerous subunits of the mediator complex as well as a variety of enzymes involved in epigenetic modification of histones.3? SR domains were found to be the determinant that facilitates B-Isox precipitation.4 Also allows for the fractionation / isolation of SG-associated proteins and RNA precipitates.5
When exposed to cells or tissue lysates, biotinylated isoxazole (b-isox) was shown to precipitate RNA-binding proteins with significant overlap to the components of RNA granules. By reversibly aggregating and disaggregating structures similar to RNA granules, b-isox has lent insight to polypeptides necessary for the reversible aggregation of RNA-binding proteins.
This product was previously listed as T511617.
Kato et al. (2012), Cell-free formation of rNA granules: low complexity sequence domains form dynamic fibers within hydrogels; Cell, 149 753
Han et al. (2012), Cell-free formation of RNA granules: bound RNAs identify features and components of cellular assemblies; Cell, 149 768
Kwon et al. (2013), Phosphorylation-regulated binding of RNA polymerase II to fibrous polymers of low-complexity domains; Cell, 155 1049
Kwon et al. (2014), Poly-dipeptides encoded by the C9orf72 repeats bind nucleoli, impede RNA biogenesis, and kill cells; Science, 345 1139
Panas et al. (2015), Methods for the characterization of stress granules in virus infected cells; Methods, 90 57