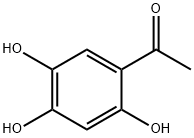
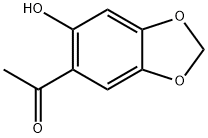
General procedure for the synthesis of 1-(2,4,5-trihydroxyphenyl)ethan-1-one from 1-(6-hydroxybenzo[D][1,3]dioxolan-5-yl)ethan-1-one: The compound 1-(6-hydroxybenzo[D][1,3]dioxolan-5-yl)ethan-1-one (4 g, 19.03 mmol) was dissolved in chlorobenzene (20 mL) at room temperature, and in a Trichlorosilane (6.6 g, 49.47 mmol) was slowly added dropwise under nitrogen protection. The reaction mixture was heated to reflux for 24 hours. Upon completion of the reaction, the chlorobenzene was removed by rotary evaporation. The residue was dissolved in 1 M hydrochloric acid (50 mL) and extracted with ethyl acetate. The extract was post-treated and purified by silica gel column chromatography to afford 1-(2,4,5-trihydroxyphenyl)ethanone (2.1 g, 12.7 mmol) as a pale yellow solid in 66.9% yield.