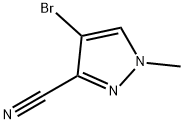
Sodium hydride (60 wt% oil dispersion, 700 mg, 17.5 mmol) was added batchwise to a solution of 4-bromo-1H-pyrazole-3-carbonitrile (2.0 g, 11.6 mmol) dissolved in 15 mL of anhydrous DMF under the protection of nitrogen, keeping the reaction temperature at 0°C. The reaction mixture was stirred continuously for 1 hr at 0°C. Subsequently, iodomethane (0.9 mL, 14.5 mmol) was slowly added to the reaction system, stirring was continued and the reaction temperature was allowed to rise naturally to room temperature within 1 hr. The reaction mixture was poured into 100 mL of ice water and stirred for 15 min before the precipitated solid was collected by filtration and washed with 20 mL of water. The resulting solid was dried overnight under vacuum and purified by silica gel column chromatography (eluent ratio 1/9 to 1/3 ethyl acetate/hexane) to afford the target product, 4-bromo-1-methyl-1H-pyrazole-3-carbonitrile, as a white solid (1.38 g, 63% yield), and a small amount of the regioselective isomer, bromide, was also isolated (100 mg).