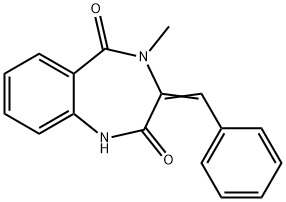
Dehydrocyclopeptine is an intermediate in the synthesis of benzodiazepine alkaloids in Penicillium. It is formed when the 3S-isomer of cyclopeptine undergoes reversible transformation by cyclopeptine dehydrogenase to displace hydrogens from the 3- and 10-positions of the benzodiazepine core.
Dehydrocyclopeptine is a benzodiazepine metabolite produced by a number of species of Penicillium. While there is little literature about the metabolite, there is considerable interest in the enzymes responsible for the biosynthesis of the benzodiazepine nucleus, cyclopeptine synthetase.
Dehydrocyclopeptine is a benzodiazepine metabolite responsible for biosynthesis of cyclopeptine synthetase. It is also used in vitro evaluation of photoprotective potential of quinolinic alkaloids isolated from antarctic marine fungus Penicillium echinulatum for topical use.
ChEBI: A 1,4-benzodiazepinone that is 3,4-dihydro-1,4-benzodiazepine-2,5-dione substituted at position 4 by a methyl group and at position 3 by a benzylidene group.